Aging Amplifies Liver Damage From Fatty Diets, Study Finds – But Rapamycin Reverses It
A new study shows aging makes the liver more vulnerable to high-fat diets, but rapamycin may reverse inflammation, restore metabolic function, and reduce fatty liver damage.
Highlights
- In mice, aging made the liver respond more severely to a high-fat diet, with more fat buildup, inflammation, and disrupted metabolic function.
- Rapamycin reversed many of those changes in older mice, including inflammatory signals and signs of fatty liver disease.
- This preclinical study suggests that aging renders the liver less resilient to dietary stress and that rapamycin may help restore some of that resilience.
As people age, the liver becomes less flexible. It is generally less efficient at handling nutrients, prone to low-grade inflammation, and vulnerable to damage from metabolic stress. This matters because metabolic dysfunction-associated steatotic liver disease, or MASLD, is becoming more common in older adults and is closely tied to obesity, poor diet, and insulin resistance. In its more inflammatory form, called MASH, the condition can progress toward scarring and even liver cancer.
A new study by Havas and colleagues, published in Aging Cell, asked a simple question. Does aging make the liver respond differently to dietary stress, and can rapamycin blunt that effect? Rapamycin is a drug that inhibits mTORC1, a nutrient-sensing pathway involved in growth, metabolism, and aging. It is already well known in aging research because it extends lifespan in multiple animal models.
Aging amplifies the liver’s inflammatory and metabolic response to a high-fat diet
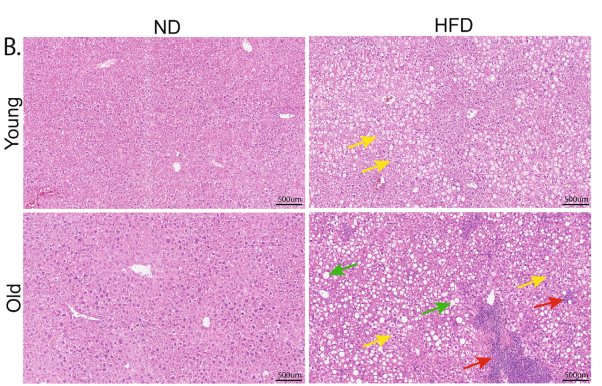
The researchers first compared young adult male mice to old male mice after nine weeks on either a normal diet or a high-fat diet. Both age groups developed signs of fatty liver disease on the high-fat diet, but the older mice showed a more severe response. Their livers had more fat accumulation, more immune cell infiltration, and more ballooning of liver cells, a sign of injury. The paper describes these changes as features of metabolic-associated steatohepatitis (MASH), including steatosis – the accumulation of excess fat in the liver.
The team then looked more closely at what was happening inside the liver. They used RNA sequencing, a method that measures which genes are more or less active, both in whole liver tissue and in isolated hepatocytes. Hepatocytes are the main working cells of the liver and handle much of its metabolic labor. Across both analyses, aging amplified the effects of the high-fat diet. Compared with young mice, old mice had stronger activation of immune and inflammatory pathways and stronger suppression of pathways involved in metabolism.
In plain terms, the older liver did not just develop more fat. It also shifted into a more inflamed and metabolically disturbed state. This is important because the combination of fat buildup and chronic inflammation is what drives the progression from simple steatosis toward more serious liver disease.
Rapamycin quiets the inflammatory response that builds up in older livers
The second part of the study focused on whether rapamycin could counter this age-amplified response. For that experiment, mice began receiving encapsulated rapamycin at four months of age. At 18 months (roughly equivalent to 50 years old in humans), some were switched to a high-fat diet for nine weeks while continuing rapamycin, creating three groups in old age: normal diet, high-fat diet, and high-fat diet plus rapamycin.
Rapamycin clearly hit its intended target. The researchers found reduced mTOR signaling mTOR in the liver cells of treated mice, confirming that the drug was active in the tissue of interest. More importantly, rapamycin reversed most of the liver gene activity changes caused by the high-fat diet. The correlation was strongly negative, meaning that many of the gene activity shifts caused by the diet moved back in the opposite direction with treatment.
A large share of the genes turned on by the high-fat diet were turned back off by rapamycin. These genes were especially involved in immune-related pathways, including innate immune activation, a first-line inflammatory response. The study also highlighted reduced activity in inflammatory regulators after rapamycin treatment, suggesting the drug may help quiet the inflammatory response that builds up in the liver under metabolic stress.
This matters because these inflammatory responses actively drive ongoing stress in the tissue, rather than simply reflecting it. The findings suggest rapamycin helps shift the liver away from this more inflammation-heavy state.
Rapamycin restores key metabolic pathways involved in fat and cholesterol processing
The researchers also found the opposite pattern. Many genes that had been suppressed by the high-fat diet were restored by rapamycin. These included genes involved in fatty acid metabolism, cholesterol metabolism, and the endoplasmic reticulum stress response. Endoplasmic reticulum stress refers to strain on the cell’s protein-folding machinery, which often rises during metabolic disease. In rapamycin-treated mice, the activity of these pathways looked much closer to that seen in mice on a normal diet.
This is one of the more important parts of the study because it shows both sides of the effect. Rapamycin lowered inflammatory activity, but it also restored metabolic programs that the high-fat diet had pushed down. In other words, the drug appeared to calm harmful stress signals while helping the liver recover some of its normal working state.
Older mice on rapamycin show less fat buildup and signs of liver damage
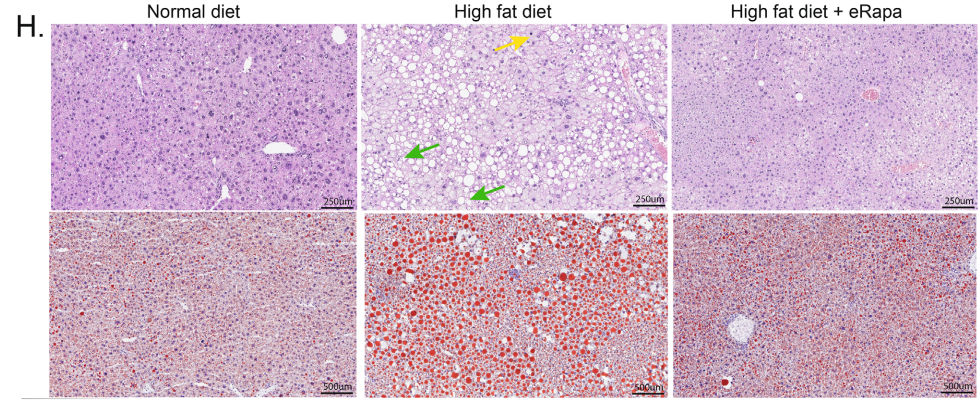
The molecular changes were matched by visible improvements in the liver. Old mice on the high-fat diet alone became much heavier than those on a normal diet. Their median body weight reached 57.8 grams, compared with 35.3 grams in the normal-diet group. With rapamycin, the median body weight on the high-fat diet fell to 41.3 grams. Liver weight also dropped, from 3.7 grams in the untreated high-fat diet group to 1.7 grams in the rapamycin group.
Microscope images told a similar story. Livers from untreated high-fat diet mice showed extensive lipid buildup and ballooned liver cells. Rapamycin sharply reduced this fat accumulation and shifted the liver’s structure closer to normal. The treatment also reduced a tumorigenic index, a gene-based score linked to liver cancer risk.
The findings suggest aging makes the liver more fragile – but also more treatable
This study does not show that rapamycin is ready to treat fatty liver disease in people. The work was done in male mice, not humans, and the authors note several limitations, including the specific diet design and the fact that food intake was not directly measured. Some of rapamycin’s effects may also have been partly related to weight loss rather than only a direct liver-specific mechanism.
Still, the findings are meaningful. They suggest that aging changes the liver’s baseline state in a way that makes dietary stress more damaging. They also suggest that rapamycin can reverse much of that response, at least in this animal model, by lowering inflammatory activity and restoring metabolic function. For aging biology, that adds to the idea that some age-related vulnerabilities may be modifiable. For liver disease, it points toward a broader strategy in which therapies aimed at aging itself may help reduce the burden of metabolic disease in older adults.

