Breakthrough in Heart Attack Recovery: Scientists Fine-Tune Cell Reprogramming Technology
Researchers change the fate of heart cells with cellular reprogramming — a genetic technique for making cells younger — to promote recovery from heart attack in mice.
Highlights:
- Human skin cells are more efficiently reprogrammed to become heart cells by blocking four specific genes, abbreviated AJSZ, with RNA interference.
- Blocking AJSZ combined with cell reprogramming reduces heart tissue scarring following heart attack in mice.
- Blocking AJSZ with cellular reprogramming improves heart function after heart attack by 50% more than normal cellular reprogramming.
Groundbreaking studies reveal that cells can be genetically reprogrammed to become younger. However, some cells are able to resists reprogramming via so-called cell fate stabilizers, which help to maintain the identity of cells. Thus, in pursuit of the ultimate anti-aging strategy, scientists have begun to find ways around fate stabilizers to erase cellular identity.
Researchers from Sandford Burnham Prebys in California have identified four genes called ATF71P, JUNB, SP7, and ZNF207 (AJSZ) that help cells resist reprogramming. Reported in Nature Communications, Missinato and colleagues show that human skin cells can be more effectively reprogrammed to become heart cells by blocking AJSZ. Furthermore, they use this anti-fate stabilization method to reduce heart tissue damage and improve heart function following heart attack in mice.
“Even if a person survives a heart attack, there could still be long-term damage to the heart that increases the risk of heart problems down the line,” says principal investigator Dr. Alexandre Colas. “Helping the heart heal after injury is an important medical need in its own right, but these findings also pave the way for wider applications of cell reprogramming in medicine.”
Blocking Fate Stabilizing Genes Enhances Cardiac Reprogramming
What makes our heart cells heart cells, or our neurons neurons? What determines cellular identity? This all depends on what genes are turned on or off in a given cell. For example, in a heart cell heart cell genes are turned on, whereas neuron genes are turned off. Cellular reprogramming works by changing which genes are turned on or off, changing the identity of cells. However, our cells possess fate stabilizers, which maintain the identify of each cell.
“Cellular reprogramming could, in theory, allow us to control the activity and appearance of any cell,” says Colas. “This concept has huge implications in terms of helping the body regenerate itself, but barriers to reprogramming mechanisms have prevented the science from moving from the lab to the clinic.”
Through a strict screening process, Missinato and colleagues identified the four strongest fate stabilizing genes (AJSZ). By inhibiting AJSZ in mouse cells, they improved heart cell reprogramming by 6-fold. Furthermore, in human skin cells, inhibiting AJSZ led to a 2.8-fold increase in cardiac-specific gene activation. ACTN2, a protein product of one of these cardiac-specific genes increased 3.2-fold. Structural changes were also observed, indicating the fate-stabilizing function of AJSZ across mouse and human cells.
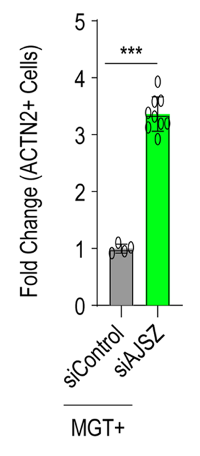
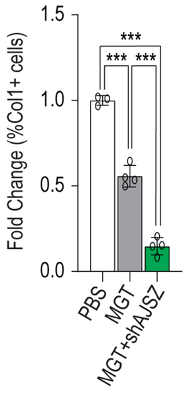
To determine whether inhibiting AJSZ could enhance heart cell reprogramming after heart attack, heart cell reprogramming genes (MGT) and AJSZ inhibitors (short hairpin RNA for each gene) were injected into the injury site of mice after heart attack (myocardial infarction). Heart attack was surgically induced by occluding the coronary artery, the main blood supply to the heart.
Twenty-five days after injection, there was a 40% reduction in heart scar tissue size and a decrease in cells positive for collagen protein. Collagen protein represents repaired tissue, as damaged heart tissue is replaced with collagen after injury, generating scar tissue. These findings suggest that inhibiting AJSZ enhances cardiac reprogramming to improve recovery from heart attack.
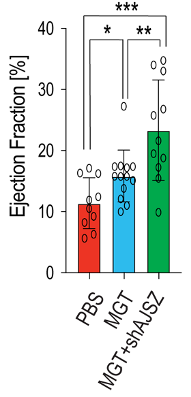
The primary purpose of the heart is to pump oxygen-rich blood to all of our cells. The volume of blood pumped from the heart with each beat — the ejection fraction (EF) is the gold standard for measuring heart function. Missinato and colleagues confirmed that the EF is reduced in mice following heart attack. Moreover, the EF improved by 100% following heart attack in mice injected with heart reprogramming genes and AJSZ inhibitors, a 50% improvement from reprogramming alone.
Cellular Reprogramming: The Ultimate Anti-Aging Technology ?
The findings of Missinato and colleagues improves on previous research showing that cellular reprogramming can improve heart function following heart attack in mice. Namely, inhibiting AJSZ improves the heart tissue regenerating effects of cellular reprogramming.
“This is helping us solve a very big problem that a lot of researchers are interested in,” says Colas. “Even more important, this breakthrough is a significant step forward on our way to turning these promising biological concepts into real treatments.”
As cellular reprogramming research continues, we may see more steps forward in the optimization of this anti-aging technique. There is certainly no lack of funding or enthusiasm for cellular reprogramming, as the CEO of OpenAI, the company behind ChatGPT, has recently invested millions of dollars into Retro Biosciences, which aims to extend human lifespan by studying cellular reprogramming.

