Can Nicotine Combat Aging? Study Links It to Higher NAD+ and Better Motor Function
New research provides evidence that long-term oral nicotine consumption raises NAD+ levels and preserves exploratory behaviors, similar to those seen during young ages, in older male mice.
Highlights
- Researchers tie the consumption of oral nicotine throughout most of the lifespan to the preservation of exploratory behaviors exhibited during younger ages in older male mice.
- An aging clock assessment based on behaviors and tissue metabolic byproduct levels suggests that nicotine makes aged male mice younger.
- Long-term nicotine consumption was also associated with gut bacteria alterations; lower levels of a gut bacteria metabolic byproduct linked to NAD+ depletion; and increased NAD+ in the muscle, pancreas, and liver.
The potential anti-aging effects of long-term nicotine use, whether from smoking, vaping, chewing tobacco, or tobacco-free oral nicotine, have remained a hotly contested topic. This comes from the two-sided nature of evidence, where, on the one hand, research has suggested cigarette smoking increases the risk of cardiovascular disease, and on the other, smoking cigarettes lowers the risk of certain age-related conditions like Parkinson’s disease. Because nicotine products have been associated with a lowered risk for some age-related conditions, a group of scientists posed the question of whether consuming nicotine without other harmful chemicals contained in cigarettes can confer effects against aging.
As published in Advanced Science, Liu and colleagues from the Chinese Academy of Sciences found that long-term oral nicotine consumption preserves the exploratory behaviors seen during younger ages in older male mice. In addition, Liu and colleagues used an age assessment based on behaviors and tissue metabolic byproduct levels, which suggested aged mice that consumed nicotine throughout most of their lives were younger. The researchers also found that nicotine consumption was associated with an altered gut bacteria composition, lower levels of ceramide (a gut bacteria metabolic byproduct tied to NAD+ degradation), and increased NAD+ in multiple tissues. These findings suggest that the long-term consumption of oral, rather than smoked or vaped, nicotine may counteract behavioral and gut bacteria signatures associated with aging, as well as aging-associated declining NAD+ levels.
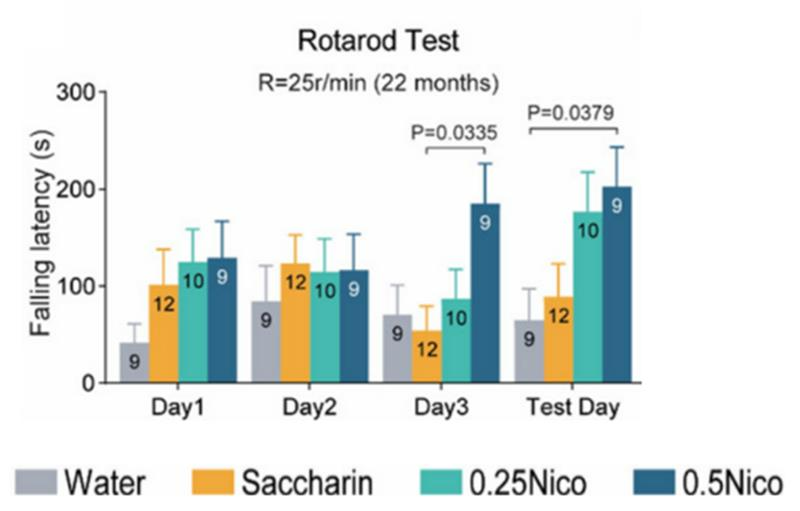
Nicotine Preserves Motor Function During Aging
Because aging is closely associated with deteriorating motor function, as observed in mice and humans, the China-based researchers performed a battery of behavioral tests to find whether nicotine might preserve motor function in mice. Interestingly, older mice given a high dose of nicotine for 22 months (roughly equivalent to 65 years for humans) exhibited enhanced coordination and endurance as well as reduced anxiety. Moreover, older mice given high doses of nicotine displayed exploratory behaviors similar to those seen in younger mice. These findings suggest that long-term nicotine consumption enhances motor function.
Nicotine Did Not Adversely Affect Cognition During Aging
To find out how taking nicotine for long periods may influence cognition, Liu and colleagues performed cognitive assessments of memory. Interestingly, nicotine treatment did not affect memory performance, suggesting that any age-related cognitive deterioration occurs regardless of whether nicotine is consumed. This finding supports that long-term usage of nicotine neither helps nor harms cognition during aging.
Nicotine Makes Mice Younger, According to an Age Assessment
To corroborate the potential anti-aging effects of nicotine, as suggested by the preservation of motor function during older ages, Liu and colleagues used an age assessment based on certain behaviors as well as metabolic byproducts in tissues. With their technique, the researchers found that aged mice treated with a high nicotine dose exhibited profiles closely resembling those of young mice. Moreover, aged mice treated with a lower nicotine dose had profiles with intermediate similarity between young and non-treated aged mice. These results suggest that long-term nicotine treatment has a dose-dependent effect on slowing aging, with higher doses conferring stronger effects.
Nicotine Increases Beneficial Gut Bacteria
Since gut bacteria composition has been linked to metabolic function (which deteriorates with age), Liu and colleagues sought to find whether long-term nicotine consumption alters gut bacteria. Notably, the researchers found that nicotine treatment, especially at higher doses, increases the ratio of beneficial to harmful gut bacteria. The ratio of beneficial to harmful bacterial species that Liu and colleagues used has been proposed as a marker of aging, and their related findings provide yet more evidence that long-term oral nicotine consumption has anti-aging effects.
Nicotine Increases NAD+ in Multiple Tissues
Because previous research has suggested that nicotine increases NAD+ synthesis, Liu and colleagues sought to determine whether long-term nicotine consumption increases tissue NAD+ levels. Interestingly, they indeed found that, at high doses, nicotine increased NAD+ levels in the liver, pancreas, and muscle, but not in fat tissue called white adipose tissue.
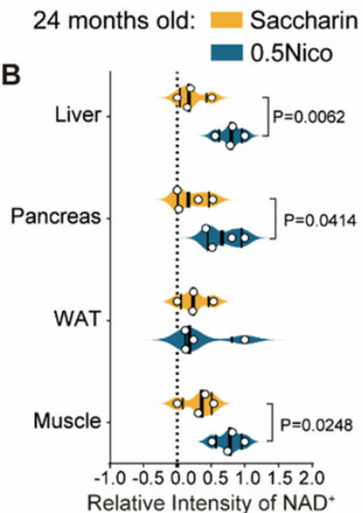
NAD+ levels have been found to decline throughout various organs and tissues in multiple organisms, including humans, during aging. Moreover, a large swath of aging researchers considers falling NAD+ levels to be a characteristic of aging. In that regard, nicotine’s effects in raising NAD+ levels offer more evidence that nicotine has anti-aging effects.
To pinpoint what underlying molecular features may contribute to increased NAD+ levels, Liu and colleagues ran correlation analyses of exploratory behaviors and gut bacteria metabolic byproduct levels in blood plasma. Intriguingly, the China-based researchers found a negative correlation between nicotine-treated aged mice that displayed behaviors similar to younger mice and the gut bacteria metabolic byproduct ceramide. In that sense, the treated older mice that exhibited younger behaviors showed associated lower levels of the gut bacteria metabolic byproduct ceramide in their blood plasma.
Ceramide has been associated with age-associated metabolic disorders and NAD+ degradation. Thus, possibly through altering gut bacteria and also lowering certain metabolites like ceramide, nicotine restores NAD+ levels and appears to rejuvenate metabolic function. By rejuvenating metabolic function, long-term nicotine consumption may also prevent an age-related deterioration in motor function as exemplified by exploratory behaviors.
“In summary, our findings reveal a previously underappreciated role for oral nicotine in promoting systemic metabolic resilience and preserving motor function during aging,” said Liu and colleagues in their publication.
Identifying Non-Addictive Compounds with Effects Similar to Nicotine
Liu and colleagues’ findings suggest that doses of nicotine, yielding comparable levels to those found in the blood plasma of moderate to heavy smokers, prevent the deterioration of motor behaviors. In that regard, if these findings apply to humans, nicotine may help preserve motor functions, such as coordination and balance, during aging. Of course, only human trials using tobacco-free oral nicotine can confirm whether this is the case, since using cigarettes would be unethical.
Furthermore, in their publication, Liu and colleagues mentioned the potential danger of administering nicotine to people for the sake of preserving motor function due to nicotine’s highly addictive properties. Nicotine’s highly addictive nature could increase the risk that people who consume oral nicotine engage in smoking or vaping, which are associated with increasing the risk of age-related conditions like heart disease. For this reason, Liu and colleagues suggested that researchers should conduct further studies to identify non-addictive compounds with molecular structures similar to nicotine, which may also increase NAD+ levels in tissues and enhance motor function during aging.
Hence, with their preclinical research demonstrating that nicotine may indeed have some anti-aging properties, Liu and colleagues may have opened the door to research exploring compounds similar to nicotine that could alleviate certain aspects of aging. In that sense, future research could unveil non-addictive compounds similar to nicotine that protect motor function as people get older, all the while avoiding the risk of age-related conditions associated with smoking.
Model: C57BL/6J male mice aged 7 weeks (roughly equivalent to 16-year-old humans) studied until age 24 months (roughly equivalent to 69-year-old humans)
Dosage: Oral nicotine in water at doses of 0.25 g/L or 0.5 g/L for 22 months

