Cell-Derived Vesicles Restore Memory and Reduce Brain Damage in Alzheimer’s, New Study Shows
Researchers from UC Irvine show that extracellular vesicles improve memory and reduce brain damage in an Alzheimer’s mouse model.
Highlights
- Stem cell–derived extracellular vesicles improve memory in a mouse model of Alzheimer’s disease.
- Treatment reduces amyloid plaques and inflammation in key brain regions.
- Benefits are observed with vesicles from both neural stem cells and lab-grown human microglia (resident immune cells of the nervous system).
We usually think of Alzheimer’s disease as one of the leading causes of cognitive decline with age. It gradually disrupts memory, thinking, and behavior, largely through the buildup of toxic protein deposits and chronic inflammation in the brain. While some treatments can slow symptoms, they do not fully address the underlying damage.
A study published in Aging Cell looks at a different way of approaching the disease. Researchers at the University of California, Irvine, investigated whether extracellular vesicles, small particles released by cells, could help restore balance in the brain. These vesicles carry proteins, RNA, and other molecules that influence how surrounding cells function.
Cell-derived signals provide an alternative to transplantation
Using extracellular vesicles instead of transplanting stem cells avoids many of the challenges associated with stem cells, including immune system rejection and safety concerns. It also shifts the focus from replacing damaged cells to modifying how existing cells communicate and function.
The researchers generated extracellular vesicles from two sources. One came from human neural stem cells. The other came from human microglia derived from induced pluripotent stem cells, which are adult cells that have been reprogrammed into a stem cell-like state and can develop into different cell types. Microglia normally help clear debris in the brain, but when overactivated, they contribute to inflammation.
These two sources represent different roles in the brain. Neural stem cells are linked to repair and regeneration, while microglia are central to immune responses and inflammation. Comparing vesicles from both allowed the researchers to see whether the therapeutic effects depended on their cellular origin.
Repeated treatment improves memory in mice modeling Alzheimer’s
To evaluate the effects of these extracellular vesicles, the researchers used 5xFAD mice, a well-established model of Alzheimer’s disease. These mice develop amyloid plaques – clumps of misfolded protein that accumulate between neurons and disrupt communication – early in life and show measurable cognitive impairment. The animals received weekly injections of extracellular vesicles over four weeks before their memory was tested with the novel object recognition test.
Healthy mice tend to spend more time exploring new objects over familiar ones, while mice with impaired memory show no clear preference. The researchers found that untreated mice modeling Alzheimer’s behaved as expected and showed little distinction between the two objects, suggesting impaired memory.
However, mice modeling Alzheimer’s treated with extracellular vesicles showed a different pattern. They spent more time exploring novel objects and performed similarly to healthy mice, suggesting an improvement in memory.
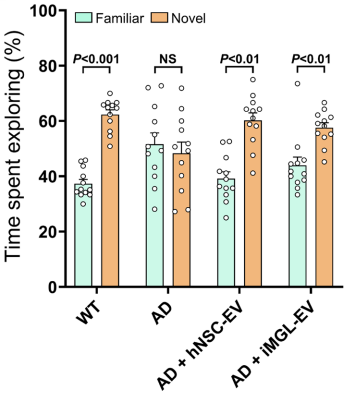
Extracellular vesicles reduce plaque buildup across brain regions
To gauge whether the extracellular vesicles affect a hallmark of Alzheimer’s, amyloid plaques, the researchers examined levels of these plaques in two regions of the brain critical for memory, the medial prefrontal cortex, which is involved in decision-making and working memory (involving both holding and manipulating information), and the perirhinal cortex, which helps recognize and distinguish familiar objects. Untreated mice showed substantial plaque accumulation in both areas.
Mice that received extracellular vesicles had significantly fewer plaques in these regions. The reduction was observed at both early and later time points, suggesting a sustained effect. Lower plaque levels are typically associated with reduced disruption of neuronal signaling, which aligns with the improvements seen in memory.
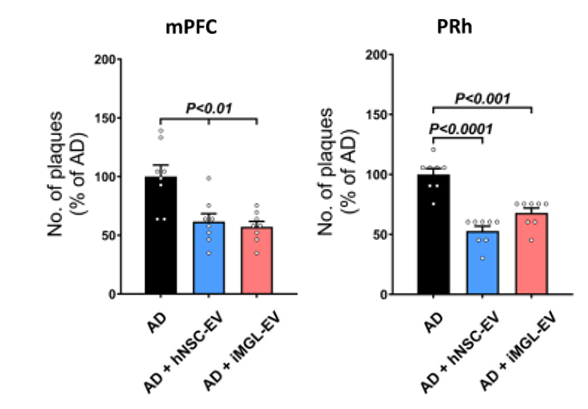
Extracellular vesicles restore synaptic function and reduce inflammation through coordinated signaling
Memory depends on synapses, the connections that allow neurons to communicate. In Alzheimer’s disease, these connections begin to break down. To assess how the extracellular vesicles affect synapses, the researchers measured two proteins associated with synaptic structure, synaptophysin and PSD-95, and found that both were reduced in untreated mice modeling Alzheimer’s, consistent with synaptic loss.
Treatment with extracellular vesicles increased the levels of these proteins, bringing them closer to those observed in healthy mice. This suggests that the treatment helps preserve or restore the connections required for memory.
At the same time, the researchers observed changes in the brain’s immune environment. Inflammation, driven in part by overactive microglia and astrocytes (cells that provide structural and metabolic support to neurons), is a central feature of Alzheimer’s disease and contributes to ongoing tissue damage. Along these lines, the researchers sought to determine whether extracellular vesicles influence microglia and astrocytes. Extracellular vesicle treatment reduced the activation of both cell types, indicating a dampening of this inflammatory response.
Together, these findings suggest that extracellular vesicles influence multiple processes at once. By restoring synaptic function while also reducing inflammation, the treatment targets two closely linked features of Alzheimer’s disease progression.
Extracellular vesicles target multiple disease pathways as clinical development begins
In this study, extracellular vesicle treatment influenced several key features of Alzheimer’s disease at once. Repeated administration reduced amyloid plaque burden, restored synaptic markers associated with neuronal communication, and improved memory in a mouse model. These effects were accompanied by reduced inflammation and changes in gene activity linked to immune signaling. Taken together, the findings suggest that extracellular vesicles act across interconnected pathways rather than targeting a single aspect of the disease.
The results remain preclinical. Translating this approach to humans will require further work to determine dosing, delivery methods, and long-term safety. Notably, more than 300 clinical trials globally are exploring extracellular vesicles, including exosomes, across conditions such as cancer, inflammatory diseases, and wound healing. Most of these studies are in early stages and are focused on safety and feasibility rather than definitive therapeutic outcomes.
Several companies are developing vesicle-based therapies, largely in regenerative medicine and immune modulation. However, clinical trials specifically targeting Alzheimer’s disease remain limited.
The growing interest in extracellular vesicles has also led to premature commercialization. Some clinics now market “exosome” or vesicle-based therapies directly to consumers, often without regulatory approval or strong clinical evidence. These treatments can vary widely in composition and quality, and in some cases may not contain well-characterized or therapeutically relevant vesicles.
At the same time, the broader concept underlying this study continues to gain traction. Many age-related diseases share overlapping biological features, including chronic inflammation, disrupted cellular communication, and impaired repair mechanisms. Approaches that influence these processes simultaneously may offer a more integrated way to address disease progression, particularly in complex conditions like Alzheimer’s.
Model: 5xFAD mouse model of Alzheimer’s disease, which develops amyloid plaque accumulation and early cognitive impairment.
Dosage: Weekly injections of extracellular vesicles derived from human neural stem cells or induced pluripotent stem cell–derived microglia administered over four weeks.

