Introducing Essential Developmental Protein Reverses Aging in Human Muscle Cells
A study shows that increasing levels of the protein NANOG can rejuvenate muscle-generating progenitor cells after they reach an aged, non-proliferating state.
Highlights
· Boosting NANOG protein levels reverses signs of aging like chromosomal instability and abnormal mitochondria by improving the DNA damage response in human muscle progenitor cells.
· Restoring muscle progenitor cells to their youthful state may offer a way to drive muscle regeneration and combat age-related musculoskeletal problems.
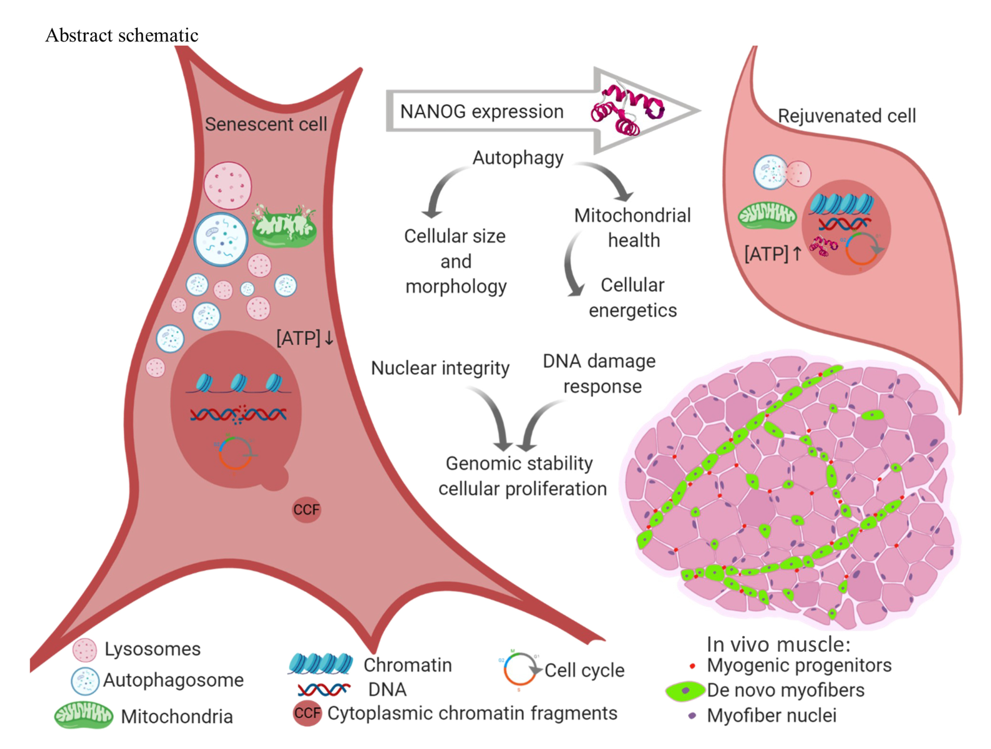
Natural aging encompasses muscle-repairing cells called myogenic progenitors entering an aged, not fully functional, and non-proliferating state called senescence. The accumulation of senescent myogenic progenitors drastically impairs muscle regeneration as we get older. With the buildup of these senescent cells impeding muscle rebuilding in all aging adults, we have a need to restore the cells’ youthful and functional state to preserve healthy muscle.
New research by Andreadis and colleagues from the University at Buffalo, NY finds that a protein called NANOG restores myogenic progenitor cell function in human cells and live mice. Published in Science Advances, their study demonstrates that this protein, named after an Irish folklore-based mythical land of youth, reverses hallmarks of cellular senescence. Such aging hallmarks include the detrimental breakup of chromosomes and the accumulation of their fragments in cells along with dysfunction of the cell’s power-generating structure, the mitochondria. In mice, NANOG restores myogenic progenitors to a functional, non-senescent state, suggesting that with just one protein, researchers can reverse skeletal muscle aging.
Essential Developmental Protein Rejuvenates Senescent Mouse Muscle Progenitor Cells
The NANOG protein has essential roles in the self-renewal of embryonic stem cells, cells crucial for development that give rise to all other types of cells in the body. And Andreadis and colleagues had previously found that NANOG restores mouse muscle progenitor cells’ potential to form new muscle, even after they reach a senescent state. Those results prompted the University at Buffalo research team to investigate whether these effects translate to human muscle progenitor cells and to fast-aging mouse skeletal muscle tissue. In their most recent study, Andreadis and colleagues showed that increasing the amount of NANOG present in cells indeed reverses hallmarks of cell senescence in muscle progenitors to restore their youthful state.
NANOG Reverses Human Muscle Progenitor Cell Aging
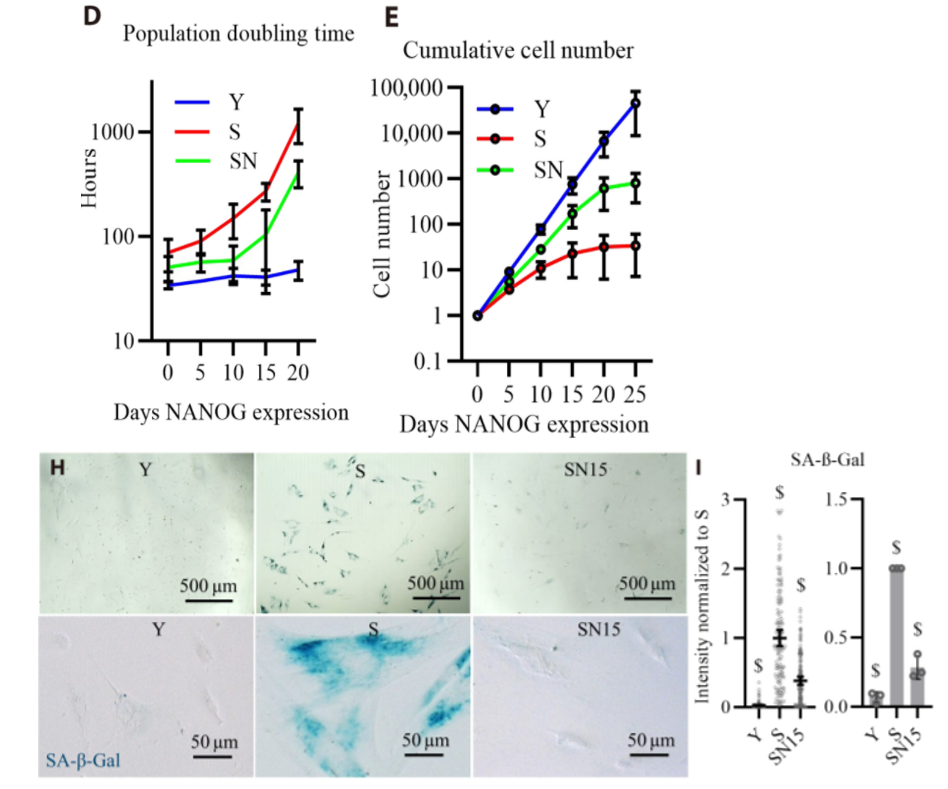
To test how elevating NANOG levels affects muscle cells, the Buffalo-based researchers treated human muscle progenitor cells with viruses to increase NANOG levels. Upon boosting NANOG levels for 5, 10, or 15 days in the senescent cells, the cells re-entered their proliferating state, increasing their numbers . What’s more, the human muscle progenitor cells showed less abundance of an aging cell marker, senescence associated β-galactosidase, indicating that NANOG reverses human muscle progenitor cell aging.
NANOG Reverses Cell Nucleus Deformities
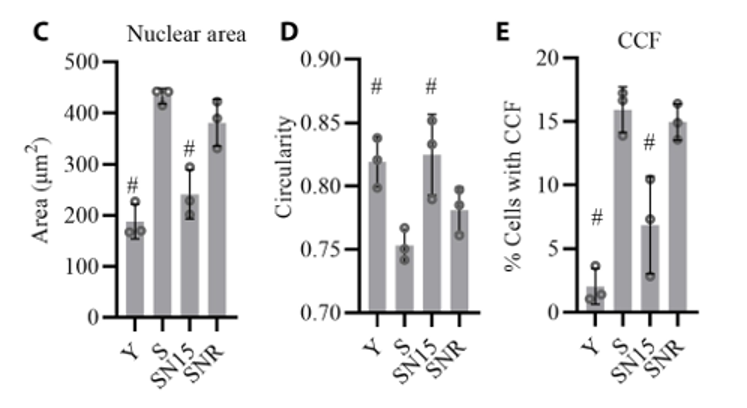
To get a closer look at how exactly NANOG rejuvenates muscle progenitors to a more youthful state, Andreadis and colleagues measured the cell nucleus size and looked for membrane deformities. They did so, because during cell senescence, the size of the nucleus enlarges and nuclear membrane deformities occur, resulting in chromosome fragments leaking to the outside of the nucleus. The researchers from the University at Buffalo found that when they increased NANOG levels in senescent muscle progenitors, the cells showed significantly decreased nuclear size. Moreover, they showed less deformities with increased nuclear circularity and a decreased presence of chromosomal fragments outside the nucleus, illustrating NANOG’s restorative effects.
Boosting NANOG Triggers A Protective DNA Damage Response
A major driver of cell senescence is the acquisition of genetic damage during aging. So, Andreadis and colleagues next sought to assess the activation of the DNA damage response, a means by which cells repair DNA. Reduced activation of the DNA damage response would indicate healthier DNA that needs less attention from the cell’s repair mechanisms. By improving the efficiency of DNA repair mechanisms like the DNA damage response to lower their activation, NANOG may revert senescent cells to their more youthful, proliferating state. And their results suggest that increasing NANOG levels triggers more efficient DNA repair with reduced activation of the DNA damage response.
Increasing NANOG Levels Improves Mitochondrial Function
Another hallmark of aging and cell senescence is the accumulation of abnormal and dysfunctional mitochondria in cells. A mechanism that disposes of malfunctioning mitochondria called autophagy can stop working properly as we age, resulting in the buildup of non-functional mitochondria. This ultimately impacts the energetic balance of cells with the accumulation of abnormal energy-producing mitochondria.
So, Andreadis and colleagues sought to find out whether improved mitochondrial health might add to the ways that increasing NANOG levels makes muscle progenitor cells more youthful. They looked at molecular markers of autophagy, the proteins Parkin and PINK1, and found that NANOG restores these proteins to youthful levels in tandem with improving membrane integrity. These results indicate that NANOG boosts autophagy in senescent muscle progenitor cells to restore and maintain mitochondrial function.
“Our work focuses on understanding the mechanisms of NANOG’s actions in hopes of discovering druggable targets in signaling or metabolic networks that mimic the anti-aging effects of NANOG,” said the study’s corresponding author Stelios T. Andreadis, PhD, in a press release.
“Ultimately, the work could help lead to new treatments or therapies that help reverse cellular senescence and aid the many people suffering from age-related disorders.”
Can Humans Use NANOG to Restore Aged Muscle Function?
We’re still a long way from figuring out whether humans can boost NANOG to alleviate age-related skeletal muscle decline. For one thing, gene therapies, like ones that would boost NANOG levels, face substantial regulatory hurdles, since they come with the potential to increase cancer risk. At the same time, the study provides valuable insight about what cellular mechanisms researchers can look at to rejuvenate skeletal muscle.
Some of the limitations from the study include that while it was shown that boosting the NANOG protein in cells facilitates a youthful state, no data was provided showing actual muscle health improvements. Also, it would be nice to see whether increasing NANOG levels increases mouse lifespan, since the true test is whether it can promote longevity. In the meantime, the wait continues to see whether potential age-reversing molecules like NANOG have any translatability to human health.