Harvard Geneticist Says We May Be in the Middle of a Longevity Revolution
Harvard geneticist George Church says reaching longevity escape velocity, where scientific advancements extending lifespan outpace life expectancy, is not out of the question by 2035.
Highlights
- Dr. George Church says he cannot rule out that humans have already reached longevity escape velocity.
- While he does not like making over-enthusiastic predictions, Dr. Church says that if we have not already reached longevity escape velocity, new technologies being researched could propel us past it by 2035.
- More specifically, key advancements that Dr. Church thinks may bring us closer to reaching longevity escape velocity include gene therapies that still need to undergo human trial testing.
Dr. George Church, a Professor at Harvard University and MIT, helped to develop the first methods for genome sequencing, as well as gene editing with CRISPR technology. He has also become a prominent figure in longevity and age-reversal research, testing the use of gene editing to reverse age-related physiological markers. In that respect, much of his current gene editing research focuses on finding ways to extend healthy human lifespan.
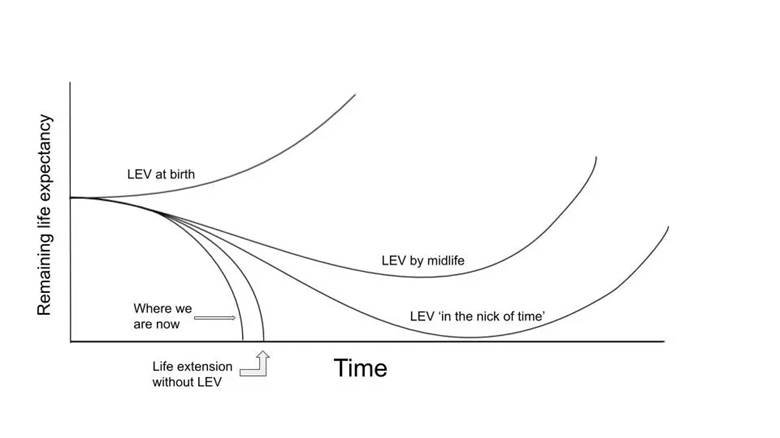
In a YouTube podcast segment, Dr. Church stated that we cannot rule out the possibility that humans have already reached longevity escape velocity (LEV), where lifespan extension from scientific advancements outpaces life expectancy. More specifically, Dr. Church said that to reach LEV, we need to add a year to lifespan each passing year, which is well below the level of detection with current measurement techniques.
Furthermore, of all of the medically related topics undergoing research, longevity is one of the most difficult to measure, according to Dr. Church. This is because measuring whether any given intervention affects human longevity would take decades to measure, since measuring longevity requires studying whether an intervention affects lifespan. To assess whether an intervention extends human lifespan, researchers would have to compare how long people live with it versus without it. Because the human lifespan can be quite long, these studies take many years. Given the difficulty of measuring longevity in humans, Dr. Church believes that when humans reach LEV, it may go mostly unnoticed, also making it nearly impossible to rule out that we have already reached it.
“I think that’s what we’re in the middle of right now, is a bunch of revolutions that people just aren’t recognizing because it’s not at the nearby drug store,” said Dr. Church. “But it is still happening.”
Along the lines of LEV, when asked if humans could reach this milestone by 2035, if indeed we have not reached it yet, Dr. Church said he does not believe it is out of the question. He also said that when LEV arrives, we may have a therapy that extends life by a year, and then, the next year, another therapy will arrive that also extends life by another year.
“One year per year is such a subtle change. Like I say, it’s lost in the noise,” said Dr. Church. “It’s going to be a broad delivery. It’s not going to be like the atomic bomb on Hiroshima. It’s going to be spread out, probably.”
Gene Therapies That May Help Humans Reach Longevity Escape Velocity
In addressing the conundrum of how to tackle aging to propel humans past LEV, Dr. Church relayed that many researchers have underestimated the complexity of aging. For example, centuries ago, explorers looked for the Fountain of Youth, a water source. Others have turned to exercise, denying certain foods, or editing one gene.
Instead of these approaches, Dr. Church thinks we will need to target multiple age-related pathways with multiple therapeutics. He continued that we will likely need to target anywhere from a dozen to hundreds of biological pathways to make substantial gains. The good news, though, according to Dr. Church, is that scientists are gaining the capacity necessary to target the complex, multifactorial nature of aging.
According to Dr. Church, some promising new gene therapy advancements may serve as the key to target multiple age-related pathways and propel us past LEV. Gene therapies are techniques that modify genes within a patient’s cells to treat or prevent aspects of aging or age-related diseases, as well as other conditions like genetic disorders. As examples, he discussed two triple gene therapies (gene therapies that target three genes simultaneously) that are designed to be secreted by the liver and then, since the liver produces about 90% of proteins in the blood, enter circulation for systemic delivery.
One of the triple gene therapies modifies the genes encoding the proteins ɑ-klotho, TGF-ꞵ receptor 2, and fibroblast growth factor 21. Scientists have used this gene therapy in various combinations to reverse age-related diseases in animal models, according to Dr. Church.
To break down the three longevity-related proteins mentioned, ɑ-klotho, a protein excreted by the kidneys, has been shown to boost cognitive function during aging in nonhuman primates. Moreover, this protein acts as a hormone to reduce cellular stress from harmful, reactive molecules, and while its levels naturally decline with age, regular exercise can support its production.
Next, TGF-ꞵ receptor 2 is a protein on the surface of cells that controls cell growth and acts as a crucial regulator of cellular aging. Some research suggests that maintaining TGF-ꞵ receptor 2 protein levels is associated with lower tumor progression in animal models of cancer and could contribute to healthier aging.
Finally, fibroblast growth factor 21 is a hormone released during states of starvation, and increasing its levels significantly extends lifespan in mice. This hormone has been shown to reduce obesity and improve blood sugar levels to potentially prevent age-related diseases.
The second triple gene therapy that Dr. Church mentioned, which he thinks holds promise, targets genes for three proteins called Yamanaka factors, which are the OCT4, SOX2, and KLF4 proteins. Yamanaka factor gene therapies have been shown to reverse aspects of aging in human cells. Also, the Yamanaka gene therapy has been shown to reverse age-related changes and extend lifespan in aged mice.
Shorter Wait Times for FDA Therapy Approval with AI
The gene therapies Dr. Church mentioned all show promise in animal models, which is the first step in getting them approved for testing safety and efficacy in human trials. In the past, it has taken therapeutics between 10 and 15 years to get through human trials and receive FDA approval. However, according to Dr. Church, researchers are using artificial intelligence (AI) to design more efficient delivery techniques for these gene therapies that make them less toxic, which could speed up FDA approval to anywhere between seven months and a few years.
It remains impossible to determine whether the gene therapies Dr. Church mentioned will prove safe and effective in humans before human trials are completed. All the same, if these therapies have similar effects in humans as they do in animal models, they could very well help us advance past LEV. Furthermore, with researchers using AI to make gene therapy delivery more efficient and less toxic, getting the FDA to approve them for humans could happen in only a few years, meaning their broad application to patients may happen in the next decade or so.
Altogether, whether humans have already developed lifespan-extending gene therapies that will drive us past LEV hinges on human trials. Interestingly, in human trials, Life Biosciences, co-founded by Harvard’s David Sinclair, is testing a gene therapy targeting genes encoding Yamanaka factors in two eye diseases, called open-angle glaucoma and non-arteritic anterior ischemic optic neuropathy. These trials aim to find whether this gene therapy can reverse these age-related eye diseases by reversing aging in neurons connecting the eye to the brain. Results from the initial trial, expected in the coming months, could provide some indication of whether gene therapies targeting Yamanaka factors work in humans relatively soon.