Longevity-Linked Protein Protects Heart Muscle From Lost Blood Flow, Study Shows
Researchers find a potential therapeutic strategy for preserving mouse heart tissue after impaired blood flow by activating a longevity-associated protein called TERT.
Highlights
· Boosting levels of the protein TERT, which is critical for telomere length maintenance, exclusively in mitochondria, protects heart muscle integrity after infarction.
· Increasing TERT levels in mitochondria enhances their power-generating capabilities to reduce dead tissue buildup after ischemic injury.
· When a procedure called RIPC, which is known to induce heart tissue protection, is performed in humans, mitochondrial TERT levels increase, suggesting translatability of these findings to people.
The loss of blood flow to the heart comes from serious events like strokes and head injuries. Paradoxically, restoring the heart’s blood supply damages muscle tissue and triggers cell death through a process called ischemia/reperfusion. The optimal functioning of heart tissue and cells is crucial for a healthy cardiovascular system and to sustain life itself. So, researchers are continuously looking for ways to protect heart tissue from injury after harmful events that disturb blood flow through this organ critical for maintaining life.
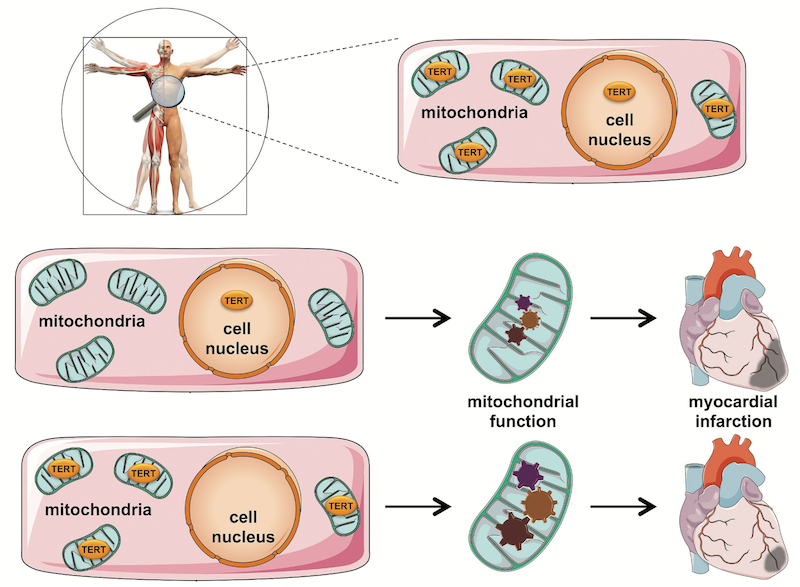
Haendeler and colleagues from the University Hospital and Heinrich Heine University in Germany published a study in Circulation showing that the longevity-linked protein telomerase reverse transcriptase (TERT) protects heart tissue after a blood flow disturbance. TERT is present in two structures within cells — the nucleus and the power-generating mitochondria — and works by extending chromosome ends called telomeres that fray and deteriorate with age. And the German research team shows that it’s the mitochondrial TERT necessary for cell energy production maintenance and heart muscle preservation. These new findings provide a potentially targetable molecular pathway for pharmaceuticals to preserve the heart following disrupted blood supply.
Mitochondrial TERT Protects Heart Tissue
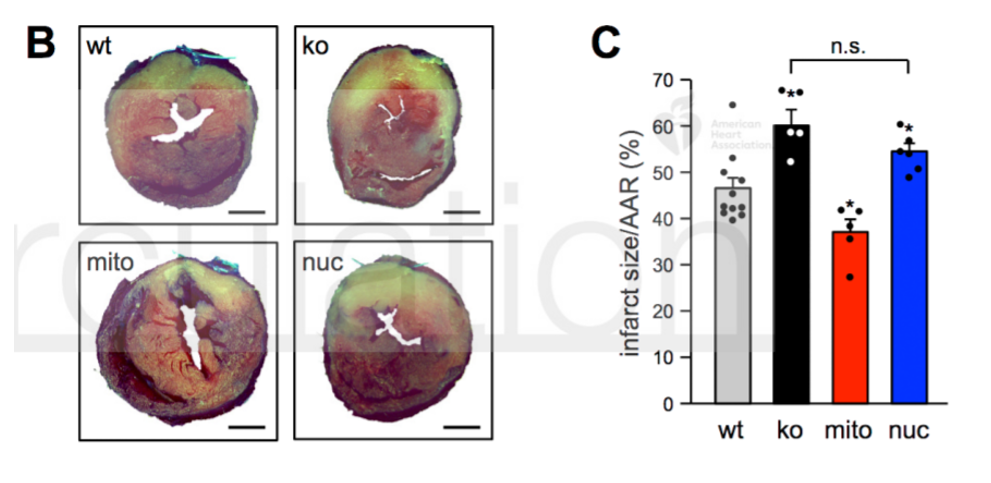
Haendeler and colleagues genetically manipulated mice to have TERT present only in the cell’s nucleus or exclusively in the cell’s mitochondria. By disrupting blood flow to the heart and restoring it, they found that mice with TERT in the nucleus only had significantly larger regions of dead heart tissue than those with TERT in the mitochondria. Intriguingly, when Haendeler and colleagues compared the amount of dead heart tissue of the mice with TERT in the mitochondria only to typical, healthy mice, they found smaller dead tissue regions with exclusively mitochondrial TERT. These results suggested that TERT in the mitochondria is critical for heart protection after blood supply disruption.
Mitochondrial TERT Improves Energy Production
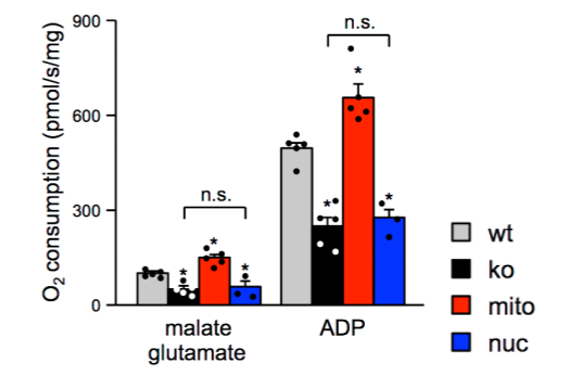
To see whether this observed tissue preservation came from improved mitochondrial function, the German research team examined TERT deficiency’s effects on mitochondrial energy production (respiration). They found that TERT exclusively in the cell’s nucleus markedly diminished respiration but that TERT in the mitochondria facilitates increased energy production. But what does the tissue preservation that arises from maintained mitochondrial energy production do for heart function after blood flow disruption?
Haendeler and colleagues next looked at the heart’s ability to pump blood after reduced blood flow and then its restoration. They found that the heart’s functional blood pumping was preserved when TERT was present only in the mitochondria. This finding points to somehow boosting mitochondrial TERT levels to preserve heart tissue and function after blood flow disruption so that when the flow is restored, less tissue death occurs. So, according to these results, increasing mitochondrial TERT levels can preserve heart function, at least in mice.
Mitochondrial TERT Protects Human Heart Tissue
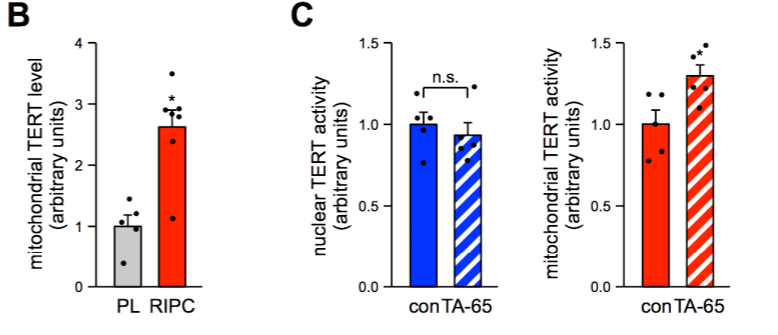
To then figure out whether this insight applies to people, Haendeler and colleagues analyzed human heart tissue from patients who underwent remote ischemic preconditioning (RIPC), an operation that can elicit heart protection. The procedure encompasses reducing blood flow for short periods before prolonged blood supply disruptions similar to stroke or head injuries. The team found that RIPC boosts mitochondrial TERT levels, which suggests that mitochondrial TERT is also crucial for heart protective effects in humans.
Haendeler and colleagues also tested the effects of the nutritional supplement TA-65, known to boost TERT levels and stimulate its function. The German team analyzed nuclei and mitochondria from heart tissue after TA-65 treatment and found higher TERT levels in only the mitochondria. These results bolstered the finding that mitochondrial TERT is responsible for TERT stimulation-related benefits in humans.
Can We Target Mitochondrial TERT to Rejuvenate Tissues?
“Using our newly created mitoTERT mice we have clearly demonstrated the cardioprotective functions for mitochondrial TERT,” said Haendeler and colleagues in their publication.
Haendeler and colleagues’ study provides optimism that we can look at the longevity protein TERT localized to the mitochondria as a molecular target to preserve cardiac tissue after serious complications like stroke. The study even examined whether their findings using mice apply to human tissue. And they found that heart protective procedures like RIPC do increase mitochondrial TERT levels in heart muscle.
Research shows that too much TERT can be a bad thing, causing cancer. So, striking the optimal levels of TERT to stimulate tissue rejuvenation while not causing cancer could have major implications for extending healthspan and lifespan. A brave and exciting new frontier of research will only tell whether manipulating TERT levels can promote tissue health and lifespan to potentially prolong the number of years we live. If TERT protein levels are what we should look to for lifespan extension, this study could be the tip of the iceberg for a new domain of longevity research.

