Max Planck Institute Scientists Show NAD+ Controls Heart Circadian Rhythm During Aging
New data show that NAD+ levels regulate the heart's circadian rhythm during aging, and the NAD+ precursor, NR, partially rescues rhythmicity and prevents heart enlargement in aged female mice.
Highlights
- Researchers show that aging disrupts gene expression patterns in the heart, which oscillate at different times of day as part of the heart’s 24-hour biological clock (known as the cardiac circadian clock), in aged female mice.
- Treatment with the NAD+ precursor NR reprograms these gene expression patterns and reverses age-related cardiac enlargement.
- In heart cells, inhibiting NAD+ synthesis adversely impacts a protein with an integral role in the cardiac circadian clock, confirming NAD+’s essential role in maintaining the clock’s rhythmicity.
The circadian clock is an internal time-keeping system that anticipates external stimuli, such as light and dark, as well as fasting and feeding cycles, and exists in nearly all mammals. Throughout the day, this clock drives rhythmic gene expression oscillations in various tissues, including cardiac tissue, which comprises the heart. These oscillations are often responsive to changes in the environment, such as those related to light, nutrients, and food.
In the heart, approximately 6% of genes are rhythmically expressed, meaning they have a role in the heart’s circadian clock (referred to as the cardiac circadian clock). Many of these genes tied to the cardiac circadian clock have been implicated in metabolic pathways, and the disruption of some of them has been shown to lead to metabolic disorders, as well as cardiovascular problems in rodents.
Increasing nicotinamide adenine dinucleotide (NAD+) with NAD+ precursor supplementation has proven beneficial in some rodent heart failure models. However, whether and how boosting NAD+ affects the cardiac circadian clock has remained unknown.
Now, as published in Communications Biology, Dierickx and colleagues from the Max Planck Institute in Germany show that the NAD+ precursor nicotinamide riboside (NR) reprograms cardiac circadian clock-associated gene expression patterns with age in mice. NR also reduced age-related heart enlargement and markers of cardiac stress. In heart cells, the researchers showed that inhibiting NAD+ synthesis adversely impacted oscillations of a protein with an integral role in the cardiac circadian clock. Collectively, these findings suggest that NAD+ precursor supplementation can restore the function of the cardiac circadian clock and alleviate heart problems during aging.
“Together, these data reveal a link between NAD+ metabolism, the circadian clock, and cardiac health during aging that could be harnessed to ameliorate cardiac defects in aged individuals,” say Dierickx and colleagues in their publication.
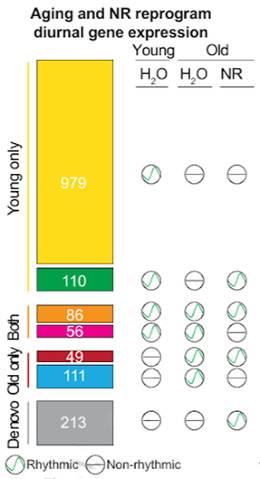
NR Reprograms Cardiac Circadian Rhythm-Associated Gene Expression
Aging has been reported to influence gene expression associated with the circadian clock in various tissues, so the researchers tested whether aging affects circadian gene expression in the heart. To do so, they measured gene expression patterns in the hearts of young, eight-week-old mice (roughly equivalent to 18-year-old humans) and older, one-year-old mice (roughly equivalent to 44-year-old humans). To assess gene expression oscillations associated with the cardiac circadian clock, they measured the gene expression at two time points, day and night.
With this protocol, the Max Planck Institute-based researchers identified 1,231 genes with oscillatory expression patterns in young mice and 302 in old ones. This showed that, similar to other organs, fewer genes exhibit oscillatory expression patterns in the hearts of old mice, which also suggests a disruption in how cardiac cells express these genes throughout the day.
To then investigate whether boosting NAD+ with NR restores the cardiac circadian clock, Dierickx and colleagues assessed gene expression at time points during the day and night after treatment with NR. The researchers began treating mice with NR at the age of two months (roughly equivalent to 18-year-old humans). The NR treatment lasted for 10 months, so that at one year old (roughly equivalent to age 44 in humans), the researchers analyzed the mice’s gene expression patterns. They took note of 213 genes that did not display gene expression oscillatory rhythmicity in young or non-treated old mice in the previous experiment, but which gained oscillatory rhythmicity upon treatment with NR. Further genetic analyses revealed that these genes, with rhythmic oscillatory expression activated by NR, code for proteins involved in longevity-regulating pathways. These results suggest that NR reprograms rhythmic oscillatory gene expression patterns in the aging heart, namely, increasing oscillatory expression of genes involved in regulating longevity.
NR Prevents Age-Related Cardiac Enlargement
Dierickx and colleagues then sought to test whether reprogramming of cardiac circadian clock-associated gene expression with NR can improve aspects of cardiac aging. Interestingly, in the NR-treated mice, cardiac enlargement, which typically occurs naturally with age, was completely prevented. Also, in line with these findings, the expression of cardiac stress marker genes was significantly reduced in older mice treated with NR.
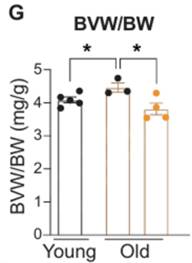
“In conclusion, our data show that NR supplementation reduces aging-induced hypertrophic growth and cardiac stress, thus revealing the therapeutic potential of NAD+ boosting during cardiac aging,” say Dierickx and colleagues in their publication.
Circadian Clock Restoration Relies on Sufficient Levels of NAD+
To confirm that NAD+ levels influence the cardiac circadian clock, the researchers tested how adding an agent, FK866, which inhibits NAD+ synthesis, to mouse heart cells affects the activity of a protein, PER2, which plays a central role in regulating the clock. Along these lines, PER2 helps to regulate the 24-hour cycle of gene expression in the heart, as well as in other tissues, such as the liver.
Indeed, treating the heart cells with FK866 reduced PER2 protein level rhythms; however, boosting NAD+ with the addition of NR restored PER2 level rhythmicity. These findings show that NAD+ levels correlate with PER2 protein level rhythms in heart cells and confirm that NR can improve PER2 protein level rhythms. Since PER2 is a critical, core component of the cardiac circadian clock, these results also suggest that NR can partially restore the clock’s function.
Figuring Out Whether NR Restores the Cardiac Circadian Clock with Age in Humans
In line with this study from Dierickx and colleagues, suggesting that NR can improve the function of the cardiac circadian clock, in human research, NR improved sleep quality and reduced drowsiness in older adults. Sleep quality relates to the circadian clock because the clock synchronizes sleep-wake cycles with the release of the sleep-promoting hormone melatonin and the wakefulness-promoting hormone cortisol.
Although the brain structure called the suprachiasmatic nucleus serves as the master regulator of the sleep-wake cycle, circadian clocks in tissues outside the brain, such as the cardiac circadian clock, also influence sleep timing and quality. For this reason, human research providing evidence that NR improves sleep quality may serve as a proxy to support that NR also helps to restore the circadian clock in tissues, such as the heart.
Some clinical trials are either completed without fully analyzed results or are in the works to test whether NR supplementation helps with heart problems. If these trials provide evidence that NR helps with certain age-related heart problems, it may also be the case that NR preserves heart function during aging by restoring the cardiac circadian clock. Only more detailed future human trials can confirm whether NR may improve heart function with age by restoring the cardiac circadian clock.
Model: Female C57Bl6J/N mice aged 2 to 4 months (young) and 12 to 15 months (old)
Dosage: NR given orally in drinking water at 3 g/L ad libitum from age 2 months to age 12 months

