NAD+ Boosting Molecule Protects Mice from Hearing Loss
Scientists find injecting mice with NAD+ booster protects against hearing loss through the activation of SIRT3, a protein that prevents damage to nerve connections between the ear and the brain.
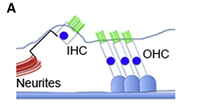
Noise exposure constitutes a major cause of hearing loss throughout the world. It causes cellular degeneration in the cochlea, a structure of the inner ear that responds to sound vibrations. In this structure, hair cells connected to nerve fibers called spiral ganglia nerve fibers, convey sound information from the cochlea to the brain.
With intense noise exposure, hair cells release neurotransmitter molecules that damage cellular connections between the cochlea and the brain. Persistent noise exposure often results in permanent damage to these structures along with permanent hearing loss.
NR Boosts NAD+ Levels to Protect Hearing
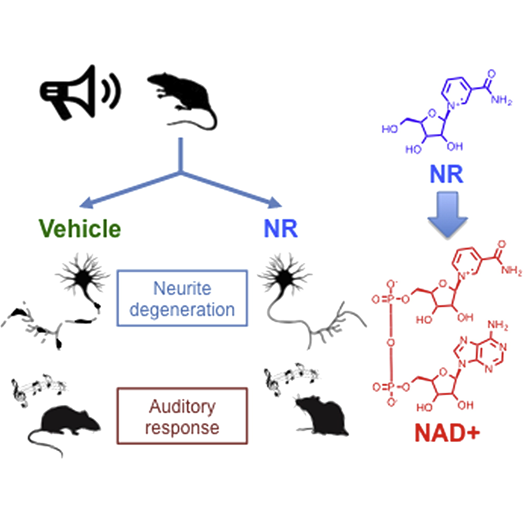
Scientists from Cornell University published a study in the journal Cell Metabolism, demonstrating that injecting mice with a molecule, nicotinamide riboside (NR), protects against hearing loss following intense noise exposure — acoustic trauma.
“This discovery identifies a unique pathway and a potential drug therapy to treat noise-induced hearing loss,” said Dr. Kevin Brown, a lead author of the study, in a press release on the study.
NR increased levels of nicotinamide adenine dinucleotide (NAD+), a molecule involved in metabolism and maintaining overall cellular health. Cells cannot readily absorb NAD+ but can absorb NR to raise the levels of NAD+ in the cell. The elevated NAD+ level from NR activates SIRT3, a protein that removes molecular tags from proteins and DNA, thereby improving cellular function. Improved cellular function in the cochlea led to protected hearing abilities following noise exposure.
Administering NR prevented decreased NAD+ levels in the cochlea of mice and protected them from hearing loss caused by noise exposure. The researchers examined SIRT3, a protein that depends on NAD+ to function, especially in the mitochondria, the cell’s powerhouse, where NR treatment increases NAD+ levels the most.
The evidence indicated that NR protected mice from hearing loss with the activation of SIRT3. Mice genetically altered to have high levels of SIRT3 showed resistance to hearing loss from acoustic trauma, which provided evidence for SIRT3 playing an essential role in preserving hearing following acoustic trauma. Genetically engineered mice without the SIRT3 protein had significantly reduced protection from hearing loss with NR treatment, showing that NR protects hearing through the activation of SIRT3.
According to the study, NR activation of SIRT3 protected the mice from hearing loss following acoustic trauma with preventing damage to nerve fiber connections to the cochlea. Moreover, organisms can absorb NR orally and the molecule reaches the cells efficiently, having “all the properties that you would expect in a medicine that could be administered to people,” said Dr. Samie Jaffrey, a contributing author for this study from Cornell University, in a statement.
At least 10 million adults under the age of 70 and perhaps as many as 40 million, or 24%, in total in the US have features of noise-induced hearing loss on their hearing tests, according to the Centers for Disease Control in a 2011-2012 study. With an aging population in the US and worldwide, identifying a therapeutic agent for noise-induced hearing loss could help improve quality of life for the aging population. Although no clinical trials are currently underway in humans to test the effects of NR on noise-induced hearing loss, future studies will examine these effects.

