NAD+ Precursors NMN and NR Suppress Hyper-Allergic Reactions
NMN and NR serve as potent therapeutics against intense allergic reactions mediated by white blood cell secretions of inflammatory factors in mice.
Highlights
- NMN and NR mitigate severe allergic reactions termed anaphylaxis as measured by diminished body temperature drops and reduced ear swelling in mice.
- Both NAD+ precursors stifle the release of proinflammatory proteins from white blood cells called mast cells.
- Genetically deleting the protein Sirt6 mitigates the benefits of NMN and NR against anaphylaxis, indicating that Sirt6 is required to suppress severe allergic responses.
Imagine going into a sudden, life-threatening allergic shock triggered by a food, drug, or chemical toxin that you may be allergic to. Research indicates that between 1.6% and 5.1% of US citizens have experienced this type of allergic response, referred to as anaphylaxis. About 1% of hospitalizations from anaphylaxis result in death, and no known treatment exists aside from an injection with epinephrine.
On a brighter note, animal research has shown that the nicotinamide adenine dinucleotide (NAD+) precursor, nicotinamide, inhibits the release of pro-inflammatory proteins called histamines from white blood cells called mast cells. Since mast cell histamine release is a hallmark of the anaphylactic response, this finding provides a glimmer of hope for boosting NAD+ to ward off these allergic reactions. However, testing the efficacy of NAD+ precursors to mitigate anaphylaxis has not been done. Why not start by testing the most potent NAD+ boosters known to date – nicotinamide mononucleotide (NMN) and nicotinamide riboside (NR)?
Published in Theranostics, Park and colleagues from Chonbuk National University Medical School in Korea showed that injecting mice with 100 mg/kg of NMN or 150 mg/kg of NR preserved the mice’s body temperature against anaphylaxis-associated temperature drops. The Korea-based researchers found that treating mice with NMN or NR significantly suppressed mast cell release of pro-inflammatory proteins after exposure to allergy-inducing molecules called antigens. The investigators also found that deleting Sirt6, essential for mast cell activation, diminished NR’s protective effects. These findings support that NMN or NR supplementation may help to quell severe anaphylactic reactions by suppressing mast cell release of pro-inflammatory proteins.
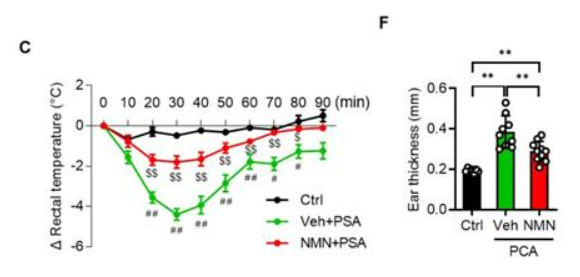
NMN and NR Supplementation Suppresses the Anaphylactic Response in Mice
To find whether NMN protects against anaphylaxis, Park and colleagues injected mice with 100 mg/kg of NMN. Twenty-four hours later, the mice were injected with anaphylaxis-inducing antigens. The research team then measured a key indicator of the anaphylactic response – body temperature – since the body’s temperature drops substantially during anaphylactic shock. The researchers saw a 4°C drop in body temperature from non-NMN treated mice but only about a 2°C drop in mice treated with NMN. Furthermore, Park and colleagues found that NMN treatment stifled ear swelling, another hallmark of the anaphylactic response. These results were reproduced with 150 mg/kg injections of NR, illustrating that NAD+ precursor supplementation can substantially mitigate the anaphylactic response in mice.
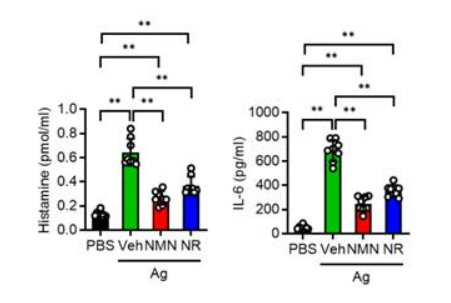
NMN and NR Dampen the Release of Proinflammatory Proteins from Mast Cells
Because mast cells play a key role in the anaphylactic response, Park and colleagues measured how NMN and NR treatment influence their release of inflammatory factors. They isolated mast cells from the bone marrow of healthy mice and treated them with allergy-causing antigens in the presence of NMN or NR. The researchers found that NMN or NR treatment reduced the release of the pro-inflammatory histone and interleukin-6 (IL-6) proteins from the mast cells by nearly half. These findings show that NMN and NR mitigate the mast cell pro-inflammatory response that propagates the physiological anaphylactic response.
The Sirt6 Protein is Required to Mitigate Anaphylaxis with NAD+ Precursors
Since Sirt6 is known to play a crucial role in regulating the mast cell response to allergens, Park and colleagues tested whether genetically deleting it would alter NMN and NR’s beneficial effects against anaphylaxis. They found that after deleting Sirt6 from mice, NMN and NR had no effect on combating anaphylaxis-induced temperature drops or ear swelling. Furthermore, neither NMN or NR could dampen the increased mast cell release of the proinflammatory proteins, histamine or IL-6. These findings indicate that Sirt6 function is necessary to quell the anaphylactic response and that activating Sirt6 could possibly provide another means to combat the anaphylactic response.
The data provided in the study seem compelling, given that NAD+ precursor supplementation nearly halved drops in body temperature and substantially decreased ear swelling during anaphylaxis. Moreover, NMN and NR also drastically reduced the amounts of histamine and IL-6 proinflammatory proteins released from mast cells in culture. The biggest question remaining is whether treating humans with NMN or NR could mitigate the effects of an acute anaphylactic shock. Since experiments have not been done in anaphylactic humans, this question remains difficult to answer. It would be hard to imagine that taking high doses of NMN or NR could stop an anaphylactic response in a person, but supplementing with one of the NAD+ precursors could, perhaps, reduce the severity of the allergic response.
Could NAD+ Precursor Supplementation Ameliorate Common Allergy Symptoms?
The findings showing that NMN or NR treatment reduces the mast cell release of the proinflammatory proteins, histamine and IL-6 begs the question of whether NAD+ precursors could help with common allergies. Anaphylaxis is on the severe end of the allergic response spectrum, and many of the same cellular processes occurring in anaphylaxis happen in common allergies. Namely, histamine and IL-6 release occur frequently and to varying degrees with common allergies. Future human trials could look at the effects of NMN or NR supplementation on blood inflammatory factor levels or examine the severity of symptoms in people with common allergies to, for example, tree pollen.

