NAD+ Supplementation Mitigates Age-Associated Metabolic Diseases like Diabetes
Boosting NAD+ levels with supplementation has the potential to protect against and mitigate metabolic diseases.
Metabolic disorders such as diabetes continue to increase worldwide with critical implications for the occurrence of life-threatening diseases, which include heart diseases, stroke, and cancer, all stemming from metabolic disorders. These disorders often come from obesity, which causes diabetes and high blood pressure, also called hypertension.
Developing metabolic disorders relates to lifestyle choices and diet. For instance, excessive energy intake and sedentary lifestyle precipitate obesity, and metabolic disorders often follow.
Replenishing NAD+ Boosts Metabolism
A review article from scientists in Japan presents how in mammalian cells, molecular pathways sensing disturbances in energy levels have self-correcting mechanisms where they increase or decrease energy levels based on biological demand. Disturbances in these molecular pathways result in metabolic disorders such as insulin resistance and fatty liver. Nicotinamide adenine dinucleotide (NAD+) acts as an energy-sensing molecule involved in these molecular pathways sensing disturbances in energy levels. This molecule plays essential roles in processes such as metabolism, aging, cell death, DNA repair, and gene expression.
Previous studies indicate NAD+ levels decline as we age and with abnormal nutrition intake, such as occurs in obesity. Decreased NAD+ levels reduce activities of NAD+-dependent enzymes involved in metabolism, along with inactivating cellular pathways of DNA repair, cellular stress responses, and metabolism regulation.
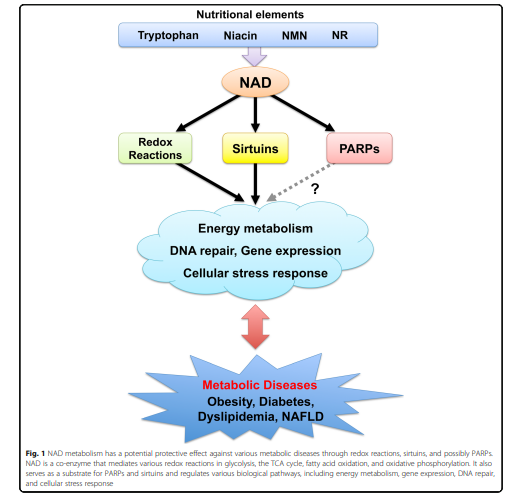
(Okabe et al., 2019 | Journal of Biomedical Science) Boosting NAD+ levels has a potential protective effect against metabolic diseases.
Studies have shown decreased NAD+ levels in numerous tissues throughout rodents with obesity. Nicotinamide mononucleotide (NMN), taken to boost NAD+ levels, can prevent this reduction in diet-induced obese mice. Moreover, mice administered NMN long-term exhibit higher physical activity with suppressed weight gain.
Obesity often leads to type 2 diabetes with insulin resistance and impaired cellular insulin secretion. NAD+ metabolism coordinates both insulin sensitivity and insulin secretion. In studies where scientists gave mice with diabetes NMN, NAD+ levels recovered, which led to the restoration of insulin secretion. Other studies have revealed that NMN administration to diabetic mice improved insulin sensitivity, increased physical activity, and promoted the function of mitochondria, the powerhouse of cells.
As studies in mice indicate nutritional intervention with NMN can improve complications from metabolic disorders, the question remains as to whether these findings can translate to humans. Scientists from Washington University School of Medicine currently run a study with oral administration of NMN to find whether this supplement can improve the function of cells secreting insulin, β-cells, in older women. Results of this study will provide evidence of whether or not NMN administration in humans can mitigate the effects of age-associated metabolic decline.

