Neuroprotective Chemicals Prevent Neurodegeneration and Preserve Function After Traumatic Brain Injury
Scientists hope that neuroprotective chemicals called P7C3 will become new interventions for nerve cell dysfunction that currently lack treatment options
On the afternoon of August 4, 2020, grey smoke with white flashes rumbled into the air at the port of Beirut. At 6:09 p.m., an explosion sent a powerful shockwave throughout the seaside city, shattering glasses, stripping away walls, exposing the rebars — skinning the buildings naked. The blast left hundreds dead and thousands injured. Many of those injured may develop traumatic brain injuries — caused by a blow or jolt to the head or even a penetrating head injury.
Blast exposure from explosives affects civilians and soldiers around the world. As the signature injury of military conflict, traumatic brain injury affects about 20 percent of the 2.3 million servicemen and women deployed since 2001. A study by researchers from the University of Iowa revealed that neuroprotective chemicals, P7C3, may prevent neuronal degeneration and preserve blast-mediated traumatic brain injury.
“It is our hope that the P7C3 family of neuroprotective chemicals will form the basis for a new class of therapeutics applicable to a variety of conditions of nerve cell dysfunction currently lacking treatment options,” writes the researchers of the study.
There are currently no FDA-approved treatments for the condition beyond supportive and rehabilitative care.
P7C3 Counteracts Traumatic Brain Injury
Traumatic brain injuries disrupt the normal function of the brain and are associated with long-term neurological complications, which may not manifest symptoms for days or weeks. However, the damage can have long-term physical, mental, and even neurological effects resembling features of Alzheimer’s disease.
The team induced traumatic brain injuries in mice by sending out a blast wave to the head and found that the animals exhibited declined memory, neuronal damage, motor coordination deficits, and signs of anxiety. The blast-injured animals showed similar symptoms as humans that suffer from traumatic brain injury.
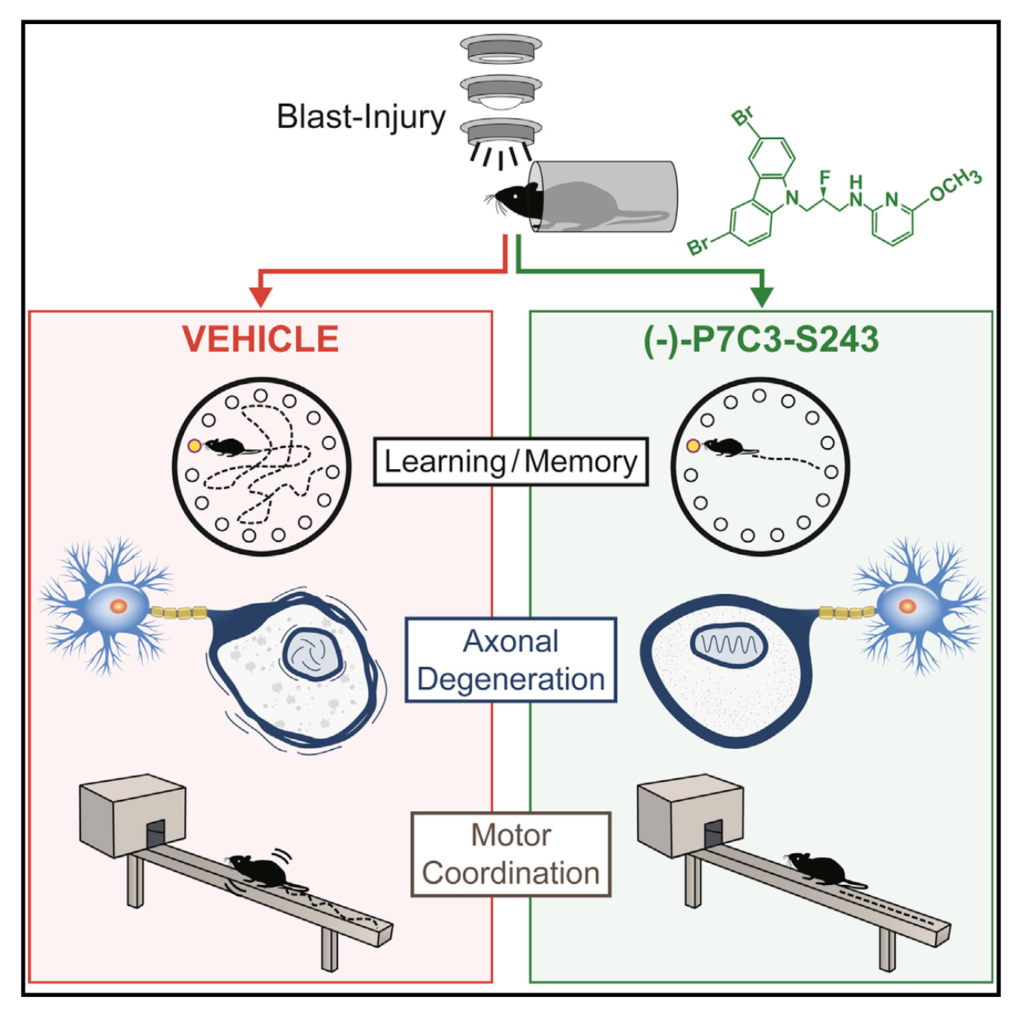
Blast exposure from explosive devices elicits traumatic brain injury associated with neurodegeneration, cognitive and motor decline, and psychiatric symptoms. The study now show that treatment with the neuroprotective agent (-)-P7C3-S243 24 hr after blast injury blocks axonal degeneration in the brain and preserves normal learning, memory, and motor coordination. Neuropsychiatric symptoms persist chronically in untreated mice. Optimized P7C3 variants offer hope for identifying neuroprotective agents for conditions involving axonal damage, neuronal cell death, or both. (Yin et al., 2014)
However, scientists from Iowa found that the neuroprotective chemicals, P7C3, can reverse some of the injury’s effects. In maze tests, while normal mice learn and memorize the route to the goal and spend about 55 percent to 60 percent of their time near the destination, blast-injured mice only spend about 25 percent, a rate achievable by simply “guessing.” But daily treatment with the neuroprotective chemical immediately after injury restored the learning and memory function to normal levels seen in uninjured mice.
Zooming in on the neurons, histologic examination of the brain tissue after the blast injury revealed a prominent neuronal degeneration similar to that observed in humans with mild traumatic brain injury. While the main body of the brain cell, where organelles reside and function, is well and alive, the communicating component, called an axon, wilts.
Anatomy of a multipolar neuron. Credit: BruceBlaus
Initiating treatment 24 hours after injury with an intermediate dose (3mg/kg/day) of oral P7C3 partially preserved axonal integrity and function. Treatment with a higher dosage (30mg/kg/day) provided full protection to the neurons. The researchers note that the protective property of P7C3 may come from its nicotinamide adenine dinucleotide (NAD+) promoting features. NAD+ is an abundant molecule in organisms that play a vital role in axon degeneration.
Moreover, the neuroprotective chemical preserved the injured animals’ motor coordination. The researchers evaluated the mice’s motor performance by counting the number of “foot slips” while the mice crossed an 80 cm (31.5 inches) beam. Twenty-eight days after the blast, the injured mice displayed a 2-fold increase in the number of foot slips, while mice with daily treatment of P7C3 were normal.
Mice with blast-mediated traumatic brain injuries also acquired anxiety-like behavior over time, correlating with increased susceptibility of humans to psychiatric symptoms after traumatic brain injury.
“Future work will focus on expanding this finding, including determination of whether acute or chronic treatment with P7C3 molecules offers protection from this aspect of [traumatic brain injuries] as well.”

