New Model Connects Mouth Microbes to Aging
An imbalance in microbes in the mouth has been linked to age-related neurological and metabolic conditions; therapeutics from traditional Chinese medicine may counteract it.
Highlights
- An imbalance in oral microbe species, characterized by a higher ratio of harmful microorganisms to beneficial ones, triggers the release of pro-inflammatory molecules into circulation.
- Harmful microorganisms from the mouth can also enter the gut through swallowed saliva, instigating systemic inflammation and age-related diseases, like diabetes and Alzheimer’s.
- Traditional Chinese medicines, such as apigenin and Zishen Wafang, can counter the release of pro-inflammatory molecules to ameliorate aspects of age-related diseases.
With an aging global population, sometimes referred to as the “silver tsunami,” researchers are constantly seeking new ways to extend lifespan spent in overall good health. In relation, as published in the Journal of Oral Microbiology, Jia and colleagues from the General Hospital of Northern Theater Command in China have proposed a new model of aging, which points to pathways to target to promote longevity.
Their model, called the oral microbiome-SASP-aging-axis, posits that an age-related imbalance in oral microbes (called oral microbiome dysbiosis) instigates the release of pro-inflammatory molecules into systemic circulation. Moreover, harmful microorganisms from the mouth can enter the gut through swallowed saliva, increasing pro-inflammatory molecules in the gut. Such pro-inflammatory molecules can trigger systemic inflammation and have been linked to an increased risk of age-related diseases like Alzheimer’s and diabetes.
To counteract oral microbiome dysbiosis and circulating pro-inflammatory molecules, the researchers reviewed the consumption of supplements with beneficial microorganisms (probiotics). Jia and colleagues also give a breakdown of traditional Chinese medicines, such as apigenin and Zishen Wafang, that can counteract pro-inflammatory molecules resulting from oral microbiome dysbiosis. These strategies may serve as ways to ameliorate the aging-associated effects of oral microbiome dysbiosis.
Details Behind Oral Microbiome Dysbiosis and Its Inflammation-Promoting Properties
The mouth houses the second-largest microbial community in the human body, only surpassed in size by the quantity of microbes in the gut. As such, microbes in the mouth are highly diverse and include bacteria, fungi, and viruses.
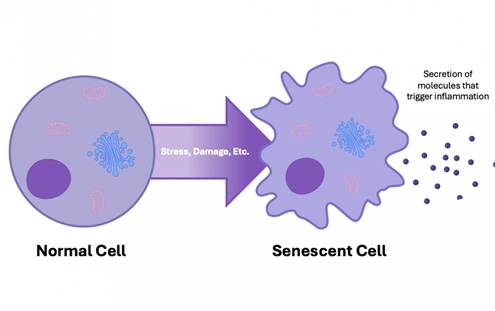
When oral microbiome dysbiosis ensues, it can accelerate the progression of age-related diseases through stimulating the release of pro-inflammatory molecules from senescent cells (dysfunctional cells that accumulate with age). The release of these pro-inflammatory molecules also drives chronic, systemic inflammation, a hallmark of aging. However, the intricate pathways behind how oral microbiome dysbiosis impacts pro-inflammatory molecule release and aging remain unclear.
Accordingly, rather than exploring exactly how oral microbiome dysbiosis may drive aging, Jia and colleagues summarized evidence that this imbalance between oral microbes is correlated with age-related diseases. Additionally, the researchers reviewed potential ways to counteract oral microbiome dysbiosis and the associated pro-inflammatory molecules linked to increased risks for age-related diseases.
Age-Related Diseases Linked to Oral Microbiome Dysbiosis
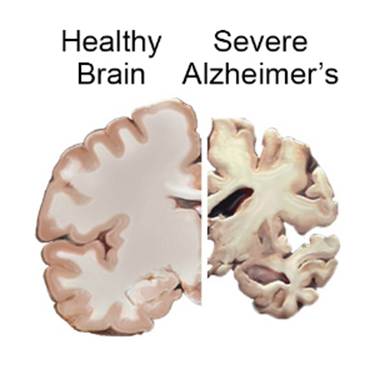
Alzheimer’s Disease
Alzheimer’s disease is a neurodegenerative condition, characterized by pathological features closely related to aging. Importantly, the progression of Alzheimer’s disease is also associated with changes in the oral microbiome.
For example, in some cases of dysbiosis, the pathogenic gum bacterium, P. gingivalis, enters the bloodstream through the use of the mouth, such as during chewing. After entering circulation, P. gingivalis crosses into the brain and drives the progression of Alzheimer’s disease features by directly damaging nerves and inducing inflammation. Since P. gingivalis increases in abundance during oral microbiome dysbiosis, its entrance into the brain and nervous system may be a way that oral microbiome dysbiosis increases the risk for Alzheimer’s.
Diabetes
Diabetes is an age-related, chronic metabolic condition where cells develop insulin resistance, causing sugar to accumulate in the blood. Some research has shown higher portions of harmful oral microbes in the mouths of elderly patients with diabetes.
As an example, studies have shown that the harmful oral bacterium, Firmicutes, is the most abundant oral bacterium in elderly patients with diabetes. Meanwhile, for younger adults, a more typical bacterium, Proteobacteria, is most abundant among oral bacterial species. Additionally, other research has comprehensively associated oral microbiome dysbiosis with diabetes. Such research associating oral microbiome dysbiosis with diabetes supports the notion that harmful microbes in the mouth contribute, in part, to this age-related condition.
Ways to Counter Oral Microbiome Dysbiosis and Associated Circulating Pro-Inflammatory Molecules
Probiotics
Taking probiotics may serve as a way to alleviate the release of pro-inflammatory molecules into systemic circulation. Corroborating this notion, supplementation with the probiotic L. plantarum has been shown to reduce the accumulation of senescent cells in bone tissue. Because senescent cells release pro-inflammatory molecules, reducing senescent cells with this probiotic may serve as a way to reduce systemic inflammation and counteract aging.
Traditional Chinese Medicine
Recent studies suggest that certain traditional Chinese medicines can effectively regulate the release of pro-inflammatory molecules from senescent cells. In doing so, they could potentially intervene against the progression of age-related diseases tied to oral microbiome dysbiosis.
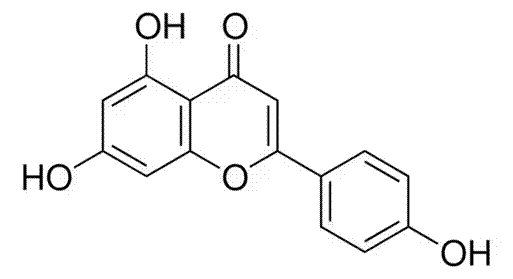
As an example, apigenin, a plant-derived antioxidant, has been shown to work as a senolytic—an agent that selectively eliminates senescent cells. In doing so, apigenin can help to reduce levels of circulating pro-inflammatory molecules.
Another traditional Chinese medicine agent, Zishen Wanfang, is an herbal extract meant to treat cognitive impairment in metabolic conditions. Research in mice has shown that this herbal extract counters blood vessel cell senescence and pro-inflammatory molecule release, thereby lowering systemic inflammation and counteracting diabetes-induced cognitive impairment. If this research translates to humans, Zishen Wanfang may help to preserve cognition in humans with diabetes. However, future human trials will be necessary for confirmation.
More Research Necessary to Prove Causation Between Oral Microbiome Dysbiosis and Age-Related Diseases
Since only correlational evidence exists between oral microbiome dysbiosis and age-related diseases, such as Alzheimer’s and diabetes, more detailed studies demonstrating causation will need to be conducted in the future. In the meantime, it is always a good idea to prevent oral microbiome dysbiosis with proper oral hygiene, limiting sugar intake, consuming fiber-rich foods, and undergoing regular dental check-ups. If more detailed research reveals that oral microbiome dysbiosis indeed increases the risk of age-related diseases and accelerates aging, it may become increasingly evident that taking care of oral health has far-reaching benefits for longevity. Once oral microbiome dysbiosis has progressed, using therapeutics like probiotics and Zishen Wanfang may also help alleviate any associated systemic inflammation.