New Study: Immune-Stimulating Therapy Reduces Body Fat and Enhances Muscles
A new therapy elicits an immune response against the muscle growth-restricting protein myostatin, reduces age-associated weight gain, and enhances muscle growth in mice.
Highlights
- The relatively inexpensive therapy, called MS2.87-97, elicited an immune response against myostatin proteins.
- MS2.87-97 treatment significantly reduced age-associated weight gain and body fat.
- MS2.87-97 treatment increased muscle mass and improved grip strength.
Myostatin is a protein produced by muscle cells that limits muscle mass by preventing excessive proliferation of muscle stem cells. In that respect, its key function is to ensure muscles do not grow too large. Due to its role in limiting muscle growth, some scientists have designated myostatin a key therapeutic target for treating an age-associated loss of muscle mass, strength, and function (a condition known as sarcopenia).
Interestingly, genetically engineered mice that have lower myostatin production display increased muscle volume and strength, as well as enhanced longevity. These findings suggest that blocking myostatin signaling could counteract age-related sarcopenia.
Now, as published in a non-peer-reviewed preprint, scientists from the University of New Mexico have shown that a therapy called MS2.87-97, which stimulates an immune response against myostatin, partially blocking its signaling, reduces age-related weight gain in mice. Moreover, MS2.87-97 reduced body fat, increased muscle mass, and enhanced grip strength. These findings support the development of MS2.87-97 as a therapy to treat obesity and age-related sarcopenia.
“If successfully brought to the clinic as an anti-obesity treatment, MS2.87-97 (or next generation anti-[myostatin] immunotherapies) could inexpensively lower non-communicable disease risk and give back years of healthy living to a hundred million Americans,” say the University of New Mexico scientists in their preprint.
Why MS2.87-97’s Development Matters
Several pharmaceutical companies have developed antibodies (proteins produced by immune cells that identify and neutralize pathogens like viruses and bacteria) to target myostatin or other proteins involved in myostatin signaling. These antibody therapies have drawbacks, though, such as requiring frequent injections, being costly, and a potential loss of efficacy over time due to the immune system eventually recognizing them and responding against them.
To get around these drawbacks, the researchers at the University of New Mexico developed a replacement for antibodies targeting myostatin. That replacement, MS2.87-97, consists of non-infectious microscopic particles that closely mimic the structure and shape of a virus and have a region on their surface similar to one found on myostatin. Through an injection of MS2.87-97 and its presentation of this microscopic region, immune cells produce antibodies against myostatin. This immune response allows antibodies to neutralize some myostatin proteins, potentially enhancing muscle growth.
MS2.87-97 Specifically Targets Myostatin
Myostatin has a high similarity to another protein called GDF11, which can play a role in bone formation. This presents a challenge in developing an immune-based therapeutic that elicits a response against myostatin, since this kind of therapeutic can also produce an off-target response against GDF11.
Along these lines, some research has shown that promoting both the degradation of myostatin and GDF11 increased muscle mass (from inhibiting myostatin) but also reduced bone density (from inhibiting GDF11) in mice. This scenario led to an increased incidence of spontaneous bone fractures, also. These findings also underscore the importance of selectively targeting myostatin without also targeting GDF11.
To ensure that MS2.87-97 specifically targets myostatin and not GDF11, the New Mexico-based scientists selected a sequence of protein building blocks (known as a peptide) from the myostatin protein that is different from any peptides on GDF11. The researchers then generated microscopic particles that have this sequence on their surface for presentation to immune cells with their MS2.87-97 therapy.
To confirm the specificity of MS2.87-97 to target myostatin and not GDF11, the scientists injected mice with the therapeutic four times, over a span of 27 weeks. Following these injections, the researchers measured antibody responses against myostatin and GDF11. They found that the MS2.87-97 injections elicited antibodies that bind to myostatin but do not detectably bind to GDF11. This finding showed that MS2.87-97 elicits an immune response specifically against myostatin and not GDF11.
MS2.87-97 Reduces Fat and Increases Muscle Mass
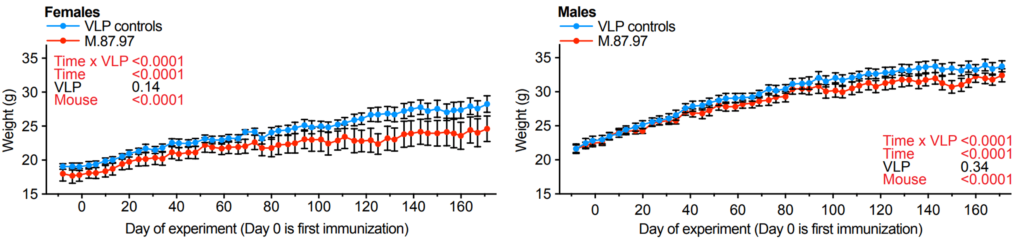
To measure whether MS2.87-97 conferred beneficial effects on aspects of physiological function, the scientists measured body weight prior to and following an injection with the microscopic particles. According to their measurements, MS2.87-97 injections were associated with significantly less weight gain over time. These data showed that MS2.87-97 attenuates age-related weight gain.
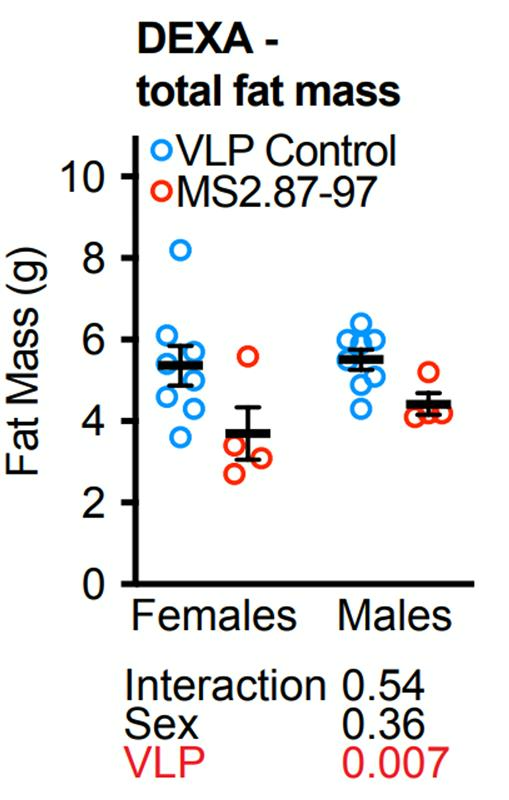
To uncover more details on what types of tissue MS2.87-97 affects to reduce age-related weight gain, the researchers utilized an assessment of body composition. With their assessment, they found that, at 4.5 months after the first injection (roughly equivalent to the age of 35 in humans), MS2.87-97-treated mice exhibited reduced total fat mass. They also found no reduction in bone mineral content, which suggested preserved bone density (another indicator that MS2.87-97 does not elicit an immune response against GDF11). These results show that attenuated age-related weight gain comes from a reduction in fat mass and that MS2.87-97 does not adversely affect bone density.
To gain insight into whether MS2.87-97 indeed mounts an immune response against myostatin to boost muscle function, the researchers assessed muscle function. At age 28 weeks (approximately equivalent to age 32 in humans), mice treated with MS2.87-97 had significantly stronger grip strength. These results showed that MS2.87-97 conferred strength gains, which may arise from suppressing myostatin and myostatin signaling.
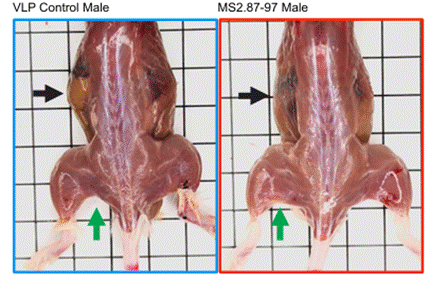
Since increased strength is often associated with increased muscle mass, the New Mexico-based scientists examined abdominal muscles, hind-end muscles, and the biceps muscles. Interestingly, they found that these three muscle groups were visibly enlarged in mice treated with MS2.87-97. These data show that, in addition to increasing strength, MS2.87-97 injections increase muscle mass.
MS2.87-97’s Potential as a New Therapeutic Option to Counteract Obesity and Age-Related Sarcopenia
In this study, University of New Mexico scientists reported the development of MS2.87-97, an injectable therapeutic containing microscopic particles that elicit an immune response against myostatin. According to their preclinical results, MS2.87-97 reduces age-related weight gain, reduces body fat, improves strength, and increases muscle mass. Future clinical trials still need to be done to confirm MS2.87-97’s benefits in humans.
If clinical trials confirm the efficacy of MS2.87-97 in humans, there are several therapeutic applications for it. One of the most obvious of these applications would be the treatment of obesity, which affects over 40% of American adults. Along these lines, age-related complications associated with obesity include diabetes, high blood pressure, cardiovascular conditions, liver disease, and some kinds of cancer.
Additionally, currently used medications for weight loss, such as Ozempic, are associated with significant loss of lean body mass, most of which is muscle instead of fat. In contrast, this study suggests that MS2.87-97 reduces fat and increases muscle mass, potentially making it comparably effective in promoting weight loss and possibly more effective in preventing age-related sarcopenia, compared to other weight loss therapies like Ozempic. Thus, if confirmed effective in clinical trials, therapeutics like MS2.87-97 could serve as affordable alternatives to popular weight loss drugs like Ozempic, without the side effect of losing lean body mass.
Model: BALB/cByJ mice
Dosage: 5 µg of MS2.87-97 virus-like particles administered via intramuscular injections at 7, 10, 16, and 27 weeks of age

