New Study Shows NMN Reduces Mortality Risk and Restores Physical Function Following Chemotherapy
The chemotherapeutic agent doxorubicin increases mortality chances, disrupts heart function, and incurs the loss of physical abilities, yet NMN counters these effects in mice.
Highlights
- NMN treatment for 10 days increases survival chances and prevents deteriorating heart function following a single high dosage of doxorubicin in mice.
- A longer-term (chronic) NMN treatment regimen also increases survival odds following doxorubicin treatments spread out over five days.
- NMN rescues physical function following the longer-term doxorubicin treatment regimen.
Doxorubicin is a chemotherapeutic agent commonly used to treat blood cancer (leukemia), lymph system cancers (Burkitt’s lymphoma and Hodgkin’s lymphoma), and ovarian cancer. Major complications from doxorubicin usage include impaired heart and physical function following treatment. Moreover, no known pharmaceuticals exist to counter doxorubicin’s adverse effects, so researchers continue to search for ways to alleviate doxorubicin-induced side effects.
Published in Cells, Grozio and colleagues from the Buck Institute for Research on Aging in California show that nicotinamide mononucleotide (NMN) improves survival odds for mice that received doxorubicin treatment. Moreover, NMN alleviated heart and overall physical dysfunction following doxorubicin treatment. The study suggests NMN may alleviate doxorubicin’s detrimental side effects.
NMN Improves Survival Odds, Heart Function, and Physical Capacity Following Chemotherapy
Since doxorubicin confers toxic cardiac side effects, the California-based researchers wanted to find whether this chemotherapeutic drives mortality in mice and if NMN could counter these effects. To study the effects of short-term treatment, Grozio and colleagues treated mice with NMN (180 mg/kg/day) injections five days before and after a single high dose of doxorubicin. In mice exposed to doxorubicin, only 48% survived, whereas NMN significantly increased survival chances to 80%. These findings indicate that NMN increases survival odds after receiving doxorubicin treatment.
Impaired heart function is a doxorubicin-induced side effect which may contribute to reduced survival odds. For this reason, Grozio and colleagues measured doxorubicin’s effects on the percentage of blood coming into the heart that gets pumped out – the ejection fraction. They found that doxorubicin treatment drastically reduced the heart’s ejection fraction, while administering NMN partially restored this indicator of heart health. These results suggest that NMN can counter doxorubicin-induced impairments in the heart’s ability to efficiently pump blood.
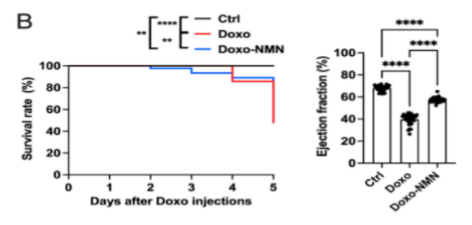
To test whether doxorubicin triggers chronic adverse effects, Grozio and colleagues treated mice with a lower dose for five days. They also administered NMN (500 mg/kg/day) in the mice’s drinking water before and after the doxorubicin injections. While only 70.4% of the untreated doxorubicin-exposed mice survived, 89.3% of the NMN-treated mice survived. These findings suggest that NMN can counter the long-term effects of chronic doxorubicin exposure on survival.
To find whether NMN can restore physical function following chronic doxorubicin treatment, Grozio and colleagues measured the maximum distances ran, maximum running times, and speed of mice on a motorized treadmill. Although doxorubicin diminished these physical function parameters, adding NMN treatment restored them to healthy mouse values. These findings suggest that NMN restores physical function following chronic doxorubicin treatment.
“We demonstrated that NMN reverses the [doxorubicin]-induced mortality and cardiotoxicity by implementing two regiments of [doxorubicin], acute and chronic,” said Grozio and colleagues. “Moreover, we showed for the first time that the oral NMN administration prevents the impairment in physical capacity that along with the onset of cardiac dysfunctions are the most common side-effects provoked by chemotherapy.”
NMN May Preserve Heart and Physical Function Following Other Chemotherapy Treatments
This study provides the first evidence that NMN can counter doxorubicin-induced physical impairments. Previous research using the chemotherapeutic cisplatin has shown that NMN can prevent kidney injury, thwart inner ear cell (cochlear cell) death, and mitigate cognitive impairment driven by cisplatin in rodents. As such, future research should examine whether NMN helps block the toxic side effects of other chemotherapeutic agents to find if it protects against all chemotherapy treatments.
Model: C57BL6/J mice
Dosage: Acute experiment – 180 mg/kg/day of NMN intraperitoneal injections; Chronic experiment – 500 mg/kg/day of NMN in drinking water

