NMN Enhances REM Sleep Brain Waves, New Study Shows
Scientists find that the NAD+ (nicotinamide adenine dinucleotide) precursor NMN (nicotinamide mononucleotide) increases the brain waves associated with REM (rapid eye movement) sleep in aged mice.
Highlights
- NMN enhances REM activity in aged mice.
- NMN appears to act on a newly discovered population of neurons.
- Genetically removing the NMN transporter reduces sleep-associated brain waves and impairs memory.
Researchers from Washington University in Missouri have found that NMN improves REM brain waves in aged mice. Their results, published in iScience, suggest that NMN may improve REM sleep by targeting a specific population of neurons in the brain.
“Our findings in this study open an interesting opportunity to further explore a potential of NMN as an effective candidate for therapeutics that can prevent or treat age-associated REM sleep problems in the elderly,” concluded the authors of the study.
NMN Enhances REM Activity
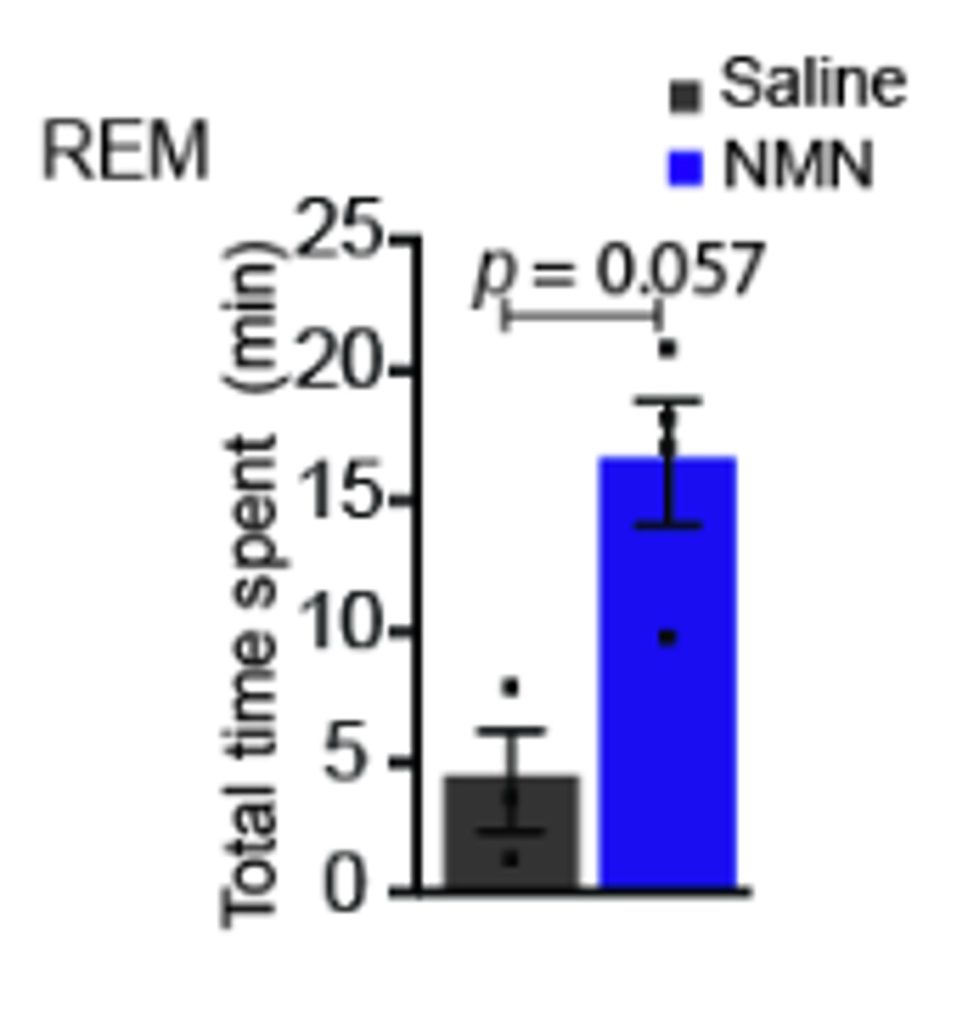
After being injected with 300 mg/kg of NMN, young and aged mice were monitored for five hours using mouse-sized EEGs (electroencephalograms). The researchers used the EEGs to measure brain wave patterns across the different stages of sleep, including wakefulness, REM, and non-REM (NREM). Strikingly, in young mice, NMN reduced wakefulness and increased both REM and NREM waves. In aged mice, NMN increased REM waves but did not significantly change wakefulness or NREM waves.
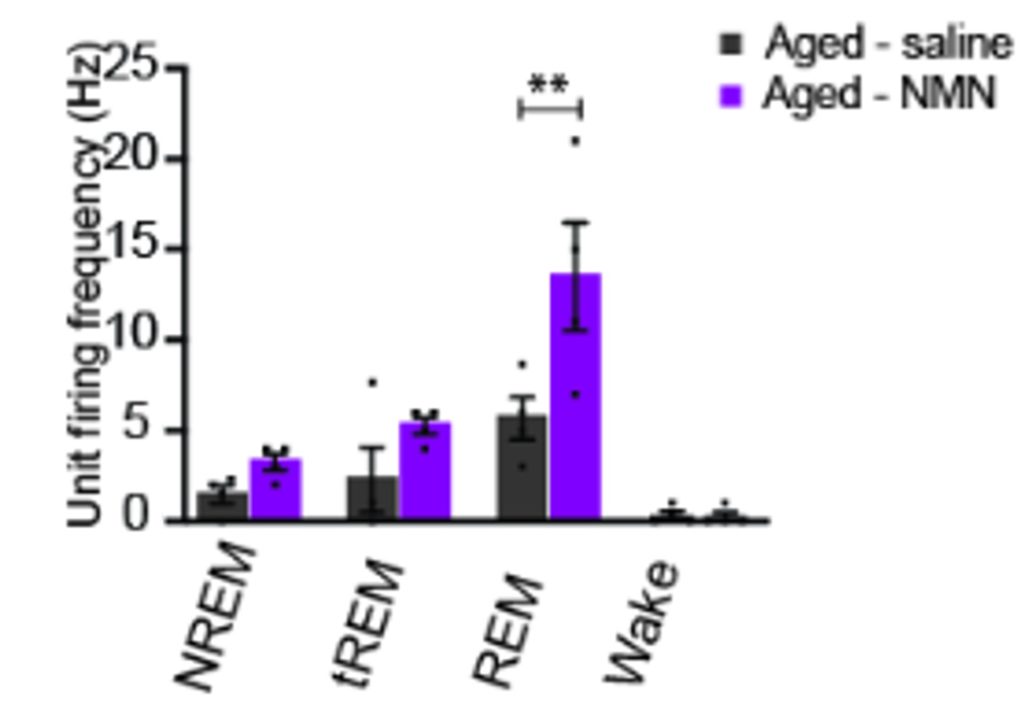
To identify which brain regions responded to NMN, the researchers used c-Fos staining, a marker of recently activated neurons. This led them to discover a previously unrecognized population of neurons in the supramammillary nucleus (SuM). They then used a carefully placed electrode to measure the electrical activity of single SuM neurons in young and aged mice. Compared with young mice, aged mice showed lower firing activity in these neurons, suggesting that their function declines with age. However, NMN increased the firing rate of SuM neurons during REM, suggesting these neurons modulate REM sleep and are sensitive to NMN.
The researchers went on to further explore whether the newly discovered population of SuM neurons regulates REM sleep. Using several approaches, they found that activating these neurons induced theta wave activity, a brain rhythm closely associated with REM sleep. In contrast, suppressing these neurons reduced REM sleep. Together, these findings suggest that this population of SuM neurons, which appears to be sensitive to NMN, contributes to REM sleep.
Removing NMN Transporter Hinders Theta Waves and Memory
The Washington University researchers, led by Dr. Shin-Ichiro Imai, were the first to identify an NMN transporter, Slc12a8, in mice. However, other researchers argue that Slc12a8 is not an NMN transporter in humans. Still, the researchers sought to determine the effect of genetically removing Slc12a8 on EEG activity. They found that theta wave activity, the brain wave activity associated with deep relaxation and light sleep, was reduced in mice lacking Slc12a8.
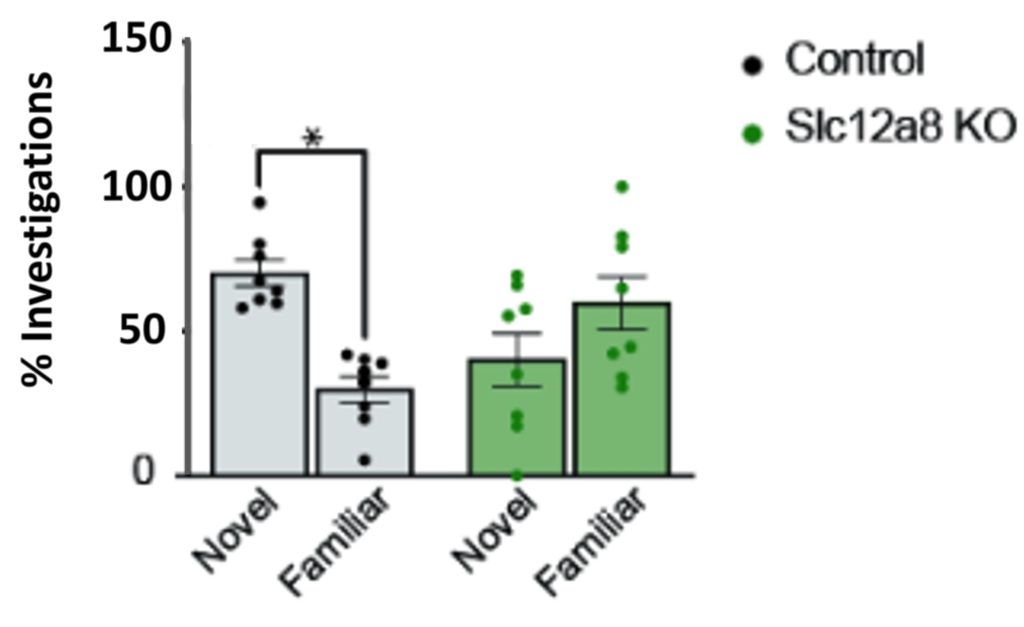
Since reduced theta wave activity during REM sleep is associated with impaired memory, the researchers tested the memory of mice lacking Slc12a8. They did so by exploiting the natural tendency of mice to explore novel objects more than familiar objects. They found that mice lacking Slc12a8 did not explore novel objects more than familiar ones, suggesting they had no memory of the familiar objects. Based on these results, the researchers presume that NMN transport through Slc12a8 is needed to induce REM sleep and maintain cognitive function.
Does NMN Improve REM Sleep in Humans?
At least two studies have shown that NMN improves perceived sleep quality in older adults. One study showed that 250 mg of NMN for 12 weeks reduced drowsiness. Another study showed that the same dosage regimen improved self-reported sleep quality. These studies suggest that NMN improves sleep in older adults, but whether it enhances REM sleep remains to be tested. While REM sleep contributes to improved sleep quality, stage 3 NREM sleep may play a more physically restorative role that leads to the feeling of being well rested.
Model: Male 20- to 24-month-old C57BL/6J mice
Dosage: 300 mg/kg of NMN administered intraperitoneally

