NMN Boosts the Efficacy of Cancer-Fighting Immune Cells
Nicotinamide mononucleotide (NMN)-treated antitumor CAR-T cells display enhanced effectiveness to combat cancer cells and reduce tumor growth in mice.
Highlights
- NMN treatment increases CAR-T cell — antitumor immune cell — proliferation up to 180 times, while prolonging the cells’ capabilities to kill cancer cells.
- With NMN treatment, anticancer CAR-T cells display signs of delayed aging as illustrated by longer telomere lengths and a reduced abundance of dead or dying CAR-T cells.
- NMN-treated CAR-T cells suppress tumor growth with longer-lasting effects in mice with cancer.
Our immune system doesn’t only seek and destroy foreign substances from outside our body, it also eliminates problematic cells internally, like cancer cells and tumor growth. However, with age our immune system becomes less efficient and internal cellular errors become more probable, leading to an increased likelihood of cancer.
In an effort to give the immune system a kick, CAR-T cell therapy has been used against malignancies, especially blood cancers, which other treatments do not effectively treat. At the same time, this treatment option is limited by poor T cell survival and lack of persistent efficacy against cancer cells. Along those lines, researchers have sought new ways to improve CAR-T cell longevity and persistent capabilities to fight against cancer.
In a non-peer reviewed preprint report, Zhong and colleagues from the Capital Medical University in Beijing China demonstrate that treating CAR-T cells with 100 µM of NMN significantly improves their efficacy and persistence to attack cancer cells. Moreover, NMN treatment enhances signs of delayed cell aging, like telomere length and reduces the buildup of dead or dying CAR-T cells. What’s more, when NMN-treated CAR-T cells were injected into mice that had experimentally-induced tumors, tumor sizes significantly diminished. These findings point to a new avenue where researchers may treat CAR-T cells with NMN to improve their viability and effectiveness to fight cancer.
NMN Treatment Enhances Cancer-Fighting Abilities
To investigate the effect of NMN on human CAR-T cell proliferation and cancer-killing capacity, CAR-T cells were treated with 100 µM NMN in the presence of NALM-6 cells in culture. NALM-6 cells are a leukemia cell line that mimic the cellular conditions of blood cancer. The China-based team found that for the NMN-treated cells, a drastic 180 times more CAR-T cells were present after 28 days of culture, compared to almost all CAR-T cells dying without NMN. Moreover, in the presence of NMN, CAR-T cells maintained their ability to kill (lyse) malignant cells longer than that of non-NMN treated CAR-T cells. These results lend credulity to the notion that NMN enhances the proliferation and tumor-killing function of CAR-T cells in culture.
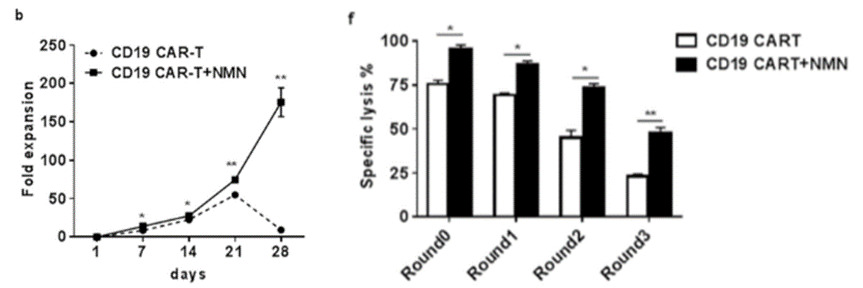
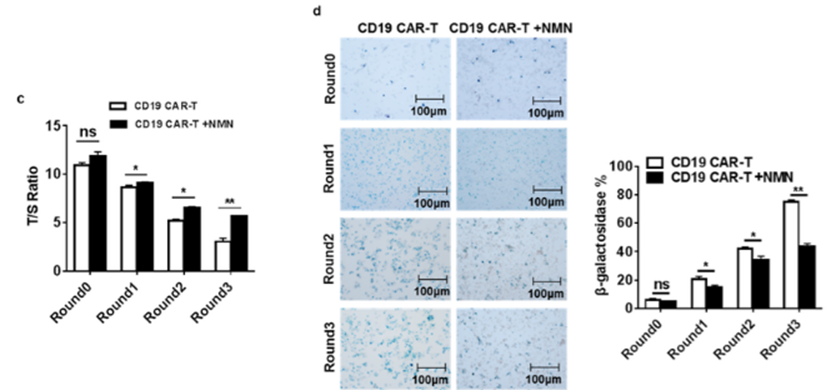
To find whether NMN delays the aging of CAR-T cells, Zhong and colleagues tested the activity of the enzyme telomerase, which extends telomeres at the ends of chromosomes. Telomerase is a critical factor in CAR-T cell replication and overall function, and its abundance diminishes in cell division, aging, and disease.With reduced telomerase activity, CAR-T cells typically die or become aged, non-functional, and non-proliferating (senescent), leading to inhibited CAR-T function and a lack of prolonged cancer-fighting capabilities. Zhong and colleagues found that treating CAR-T cells with NMN increased telomerase activity while senescent CAR-T cell abundance substantially decreased. This finding confirms that NMN extends CAR-T cell longevity and prolongs action by promoting telomerase activity and reducing the numbers of senescent CAR-T cells in culture.
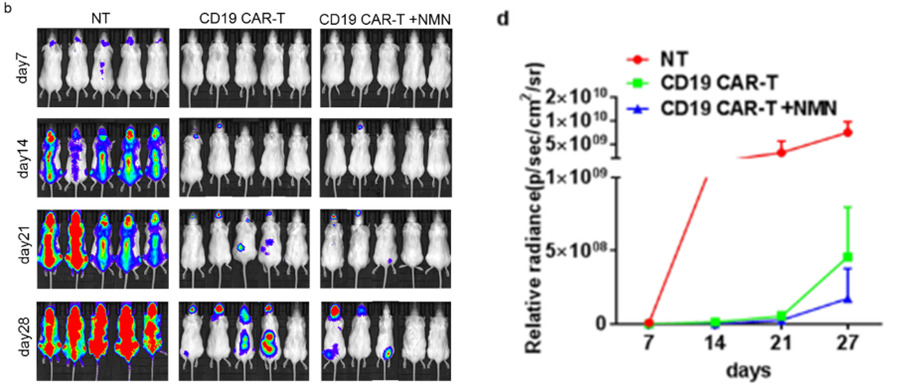
NMN-Treated CAR-T Cells Suppress Tumor Growth in Mice
Zhong and colleagues next sought to find what effects NMN-treated CAR-T cells have on tumors in mice. Mice were first injected with NALM-6 leukemia cells to generate a mouse cancer model and then injected with NMN-treated CAR-T cells. The Beijing-based team found that treatment with NMN-treated CAR-T cells promoted longer-lasting tumor suppression as shown by lower tumor volumes. These findings provide further support that NMN enhances the cancer-fighting capabilities of CAR-T cells as demonstrated in this mouse model for cancer.
NMN Stimulates the Longevity-Associated Sirt1 Gene
To find out what genes NMN stimulates, Zhong and colleagues performed gene activity analyses and found that NMN promotes the activation of the Sirt1 gene, associated with cell longevity. In NMN-treated CAR-T cells, increased Sirt1 activity was also linked with decreased activation of cell death and inflammation-associated genes. Since Sirt1 has a presumed role in suppressing the activation of cell death-associated genes, these findings provide some clues for the cell mechanism through which NMN promotes CAR-T antitumor efficacy.
Identifying Which Precise Cell Mechanisms NMN Stimulates
“In this study, we found that NMN can increase cell rejuvenation and cell proliferation by upregulating Sirt1 and provide us with great confidence NMN can enhance CD19 CAR-T cell longevity and anti-tumor efficiency,” said Zhong and colleagues in their publication.
The findings of the study highlight the potential clinical application of NMN for CAR-T cell human cancer immunotherapy. With NMN application, human CAR-T cells were more viable and had better capabilities to kill cancer cells. These findings also applied to a mouse cancer model, where injections of NMN-treated CAR-T cells reduced tumor growth, illustrating that NMN may be the key to enhance CAR-T therapy.
Future studies need to elucidate the precise cellular mechanisms by which NMN promotes CAR-T viability, longevity, and cancer-fighting abilities. A more detailed look at NMN’s effects on cells will allow researchers to pinpoint other pathways that NMN influences. In doing so, we may gain further insight into improving CAR-T cell function to fight against cancer.
This study provides insights into how NMN helps rejuvenate the immune system by jump starting CAR-T cells. Since the immune system declines with aging, it is possible that NMN could be used as a preventative measure against cancer growth.

