NMN Inhibits the Onset of Liver Fibrosis
Nicotinamide mononucleotide (NMN) injections inhibit mouse liver cells from driving scarring that leads to liver fibrosis.
When our livers get injured, whether from chronic alcohol use or the buildup of cellular stress in the form of damaging molecules called reactive oxygen species, it can cause scarring, known as fibrosis. This excessive scarring can lead to diseases like liver cancer and cirrhosis and, currently, no therapeutic options to prevent it exist.
Deng and colleagues published a study in Free Radical Biology and Medicine describing how injecting mice with nicotinamide mononucleotide (NMN) prevented liver fibrosis. The research team from Tsinghua University in Beijing found that NMN supplementation inhibited gene activity typically activated in cells that promote liver fibrosis. Also, NMN promoted the presence of the protein 15-PGDH, which reduced the levels of PGE2, a lipid that drives liver fibrosis. These results could translate to helping people prevent liver fibrosis someday.
Reduced NAD+ Levels in Liver Disease and Injury
Nicotinamide adenine dinucleotide (NAD+) is a vital molecule in cellular energy production as well as maintenance of DNA integrity. But its levels decline with age in animals and humans alike, which has been linked to the onset of age-related diseases from Alzheimer’s disease to metabolic dysfunction and liver disease. Not only that, but previous research indicates that regular alcohol consumption reduces NAD+ levels in the liver, tying reduced NAD+ levels to liver injury.
There are several precursors to NAD+ including NMN. Studies indicate that supplementing mice with NMN increases levels of NAD+ in cells. For these reasons, Deng and colleagues tested whether NMN can alleviate symptoms in liver fibrosis.
Suppresses Gene Activity Linked to Liver Fibrosis
In their study, the scientists found NMN suppressed genes that make proteins linked to liver fibrosis onset. They examined a type of cell called hepatic stellate cells, the main source of proteins causing liver fibrosis. The researchers analyzed the gene levels of liver fibrosis proteins in these cells, and after NMN treatment, the levels of genes encoding these proteins diminished. They treated these cells with an enzyme called TGF-β1 to increase the levels of genes encoding proteins secreted from cells during liver fibrosis, which was reversed with NMN treatment.
Prevented Liver Fibrosis in Live Mice
Next, the researchers wanted to find out whether these results obtained from hepatic stellate cells in a dish translated to preventing liver fibrosis in live mice. They found NMN supplementation diminished the presence of markers of tissue scarring in mice treated with a molecule that induces liver fibrosis, thioacetamide. When the scientists analyzed the liver tissue of these mice after 32 days of treatment, they found more collagen deposits in the tissue indicative of liver fibrosis, which improved with NMN supplementation.
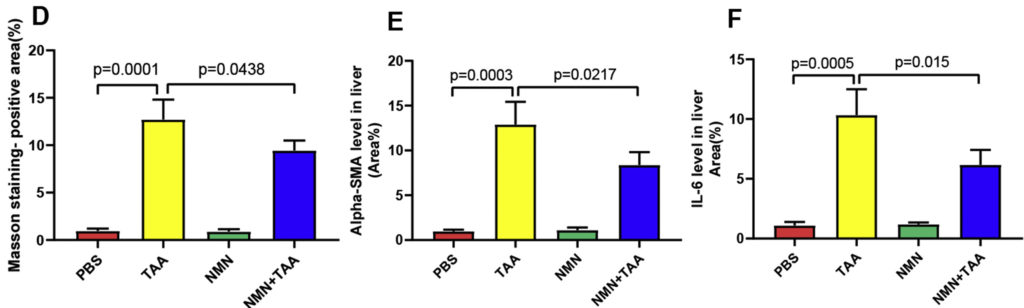
Inhibits Liver Fibrosis Onset by Elevating 15-PGDH Levels
To investigate how NMN suppressed liver fibrosis at the level of cells, the team analyzed protein changes that happen with NMN administration. They found NMN increased the amount of a protein called 15-PGDH with or without inducing liver fibrosis. Previous research had shown that 15-PGDH degrades with high cellular levels of harmful molecules called reactive oxygen species, so they proposed that NMN reduced the presence of these reactive molecules in cells. They also showed that increased levels of 15-PGDH inhibited hepatic stellate cell activation. Higher 15-PGDH levels led to reduced levels of PGE2, a lipid that promotes liver fibrosis, providing evidence for how NMN prevents liver fibrosis.
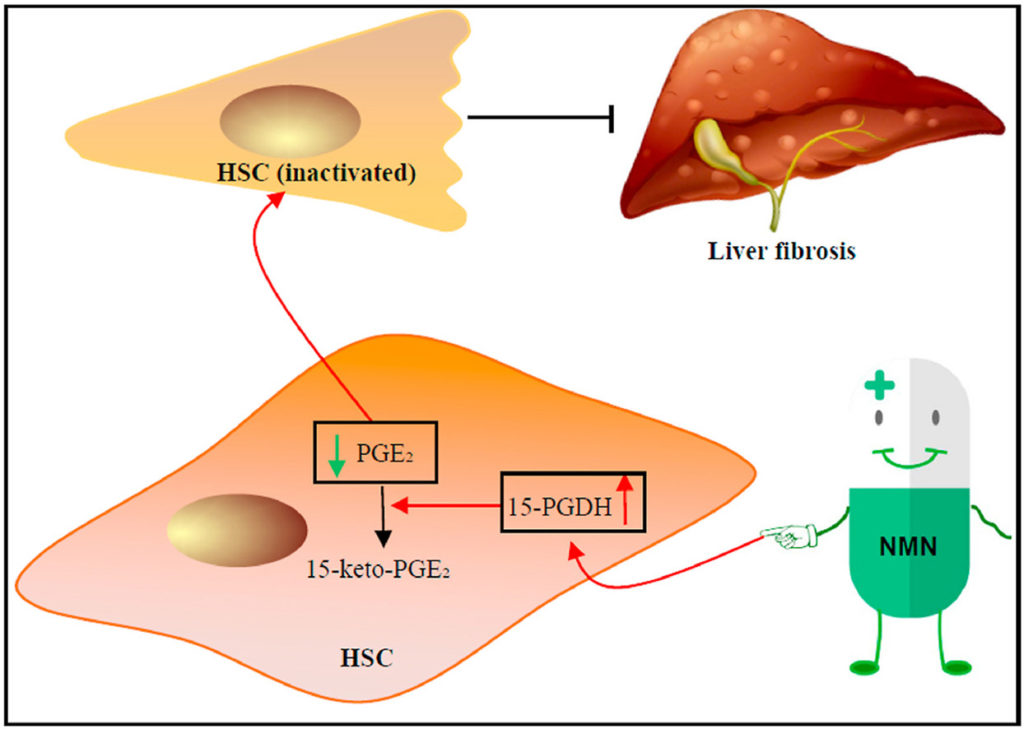
“Using mouse models for liver fibrosis, we demonstrated that NMN effectively reduced the production of extracellular matrix (ECM) and prevented liver fibrosis,” stated Deng and colleagues.
The study’s results indicate that increasing NAD+ levels with NMN can prevent liver fibrosis. This means that NMN may prevent liver fibrosis stemming from different forms of liver injury that result from the buildup of damaging reactive oxygen species. The team went on to show that NMN increases 15-PGDH levels by protecting it from being degraded by reactive oxygen species. Increased levels of 15-PGDH then break down the lipid called PGE2 that activate hepatic stellate cells, preventing liver fibrosis.
“These results propose that NMN supplementation inhibits [hepatic stellate cell] activation and prevents liver fibrosis in mice, providing a new opportunity for developing liver fibrosis therapy,” said Deng and colleagues.
Clinical trials are needed to show whether these beneficial effects on liver fibrosis will translate to people. Since liver fibrosis frequently precedes the onset of liver cancer, these beneficial effects would have significant therapeutic value.

