Japanese Study Indicates NMN Injections Reduce Blood Fat Content in Healthy Individuals
Injecting healthy individuals with NMN safely and effectively reduced blood fat (triglyceride) content and increased NAD+ levels.
Highlights
- A single injection of NMN reduces blood fat content by about 75%.
- NMN injections are safely metabolized and cause no discernible damage to the heart, kidney, or pancreas despite bypassing the body’s detoxifier – the liver.
- The injections boost blood NAD+ levels by about 20%.
Rodent studies suggest nicotinamide mononucleotide (NMN) supplementation reverses age-related diseases, and human studies show it improves insulin sensitivity and muscle function. So, as a potential preventive measure against aging, several clinics in Japan have begun to administer NMN by intravenous injection. While oral administration of NMN has been demonstrated as safe, the safety of intravenously injecting NMN has not been confirmed. When injected directly into the bloodstream, NMN bypasses the body’s central detoxifier, the liver. Concern has mounted that without liver filtration, NMN may cause damage to the heart, pancreas, and/or kidneys.
Published in Cureus, Gotoh and colleagues from Hyogo Medical University in Japan show that NMN is safely metabolized and substantially reduces blood fat (triglyceride) levels when injected. Furthermore, injecting NMN significantly raises blood levels of the pro-longevity molecule nicotinamide adenine dinucleotide (NAD+). Interestingly, no other human study to date has shown NMN reduces blood triglyceride levels, possibly because oral supplementation was used. These findings suggest that NMN injections can be safely metabolized and may help reduce blood triglyceride levels.
NMN Injections Reduce Blood Fat and Increase NAD+
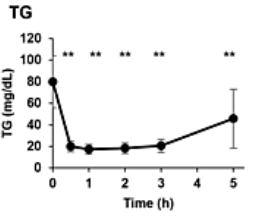
To measure whether NMN injections confer metabolic benefits, Gotoh and colleagues intravenously injected ten healthy individuals (age 20-70 years) with 300 mg of NMN and performed blood analyses. Although NMN injections had no effect on cholesterol, it reduced blood triglycerides by about 75% (from ~80 mg/dL to ~20 mg/dL after three hours). No study using oral NMN administration has demonstrated reduced triglyceride levels, suggesting a unique benefit from NMN injections. Elevated triglyceride levels have been linked to fatty liver disease and type II diabetes, so NMN injections may provide a way to counter these age-associated diseases.
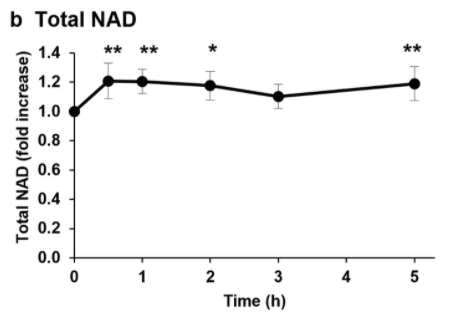
To see whether NMN’s benefits arise from increasing levels of the essential pro-longevity molecule NAD+, the Japanese researchers analyzed blood NAD+ levels. The research team found that NMN injections increase NAD+ levels by about 20% five hours after the injection. A dip in NAD+ content was observed three hours following injection, for which the research team had no plausible explanation. These findings suggest that the effects of NMN, including reduced triglyceride levels, come from boosting NAD+ levels.
“Our clinical study indicated that 300 mg NMN administration is tolerated by humans because it does not cause significant damage to blood cells, the liver, pancreas, heart, and kidneys when injected intravenously and effectively increases the amount of NAD+ in blood cells,” said Gotho and colleagues.
Injecting NMN May Confer Benefits Not Seen with Oral Use
The study of Gotho and colleagues suggests that NMN administered intravenously can be safely metabolized despite bypassing the liver. This route of administration may reap benefits that oral NMN doesn’t provide, such as reduced triglyceride levels. Future studies should examine how intravenous injection of NMN affects additional age-related conditions like muscle decline, insulin insensitivity, and reduced NAD+ levels in organs like the skin. It’s possible that NMN injections provide benefits that previous clinical trials have not detected with oral treatment. Moreover, the effects of NMN injections should be measured over longer periods.
Model: Healthy individuals aged 20 to 70 years
Dosage: Intravenous injection of 300 mg of NMN

