NMN Limits the Initiation of Precancer, According to New Study
Scientists find that NMN (nicotinamide mononucleotide) mitigates the conversion of stomach cells into intestinal-like cells, which may potentially reduce the risk of stomach cancer.
Highlights
- Researchers developed a miniature organ called an organoid to more accurately model a precancerous condition called gastric intestinal metaplasia (GIM).
- Low NAD+ levels were found to contribute to the cellular changes that occur with GIM in human tissue, the organoids, and four different mouse models.
- NMN mitigated GIM in the organoid model, as well as four different mouse models.
Stomach cancer, the fifth most common cancer in the world, often leads to early mortality, thereby blunting longevity. Before stomach cancer develops, stomach cells can lose their identity and become intestinal-like cells. Scientists have named this mosaic of cells gastric intestinal metaplasia (GIM). Notably, studies have shown that reversing GIM can prevent the progression of stomach cancer. Now, Zhengzhou University scientists have found that NMN may reverse GIM, as published in a new study.
Low NAD+ (Nicotinamide Adenine Dinucleotide) Levels in Metaplasia
Adequately recapitulating human GIM with animal models has challenged scientists for years. However, the last few decades have brought organoids to the labs of many researchers. Organoids are miniature organs that model the cellular complexity of actual organs. They can more accurately model the cellular changes that occur with GIM.
To model GIM, researchers artificially hyperactivated CDX2, a master gene regulator, within stomach organoids. CDX2 controls genes that are essential for intestinal cell development. Thus, within the stomach organoids, CDX2 hyperactivation converted many stomach cells into intestinal-like cells, thereby recapitulating GIM. The resulting GIM organoids featured crypts, which are structures that belong in the intestines but not the stomach.
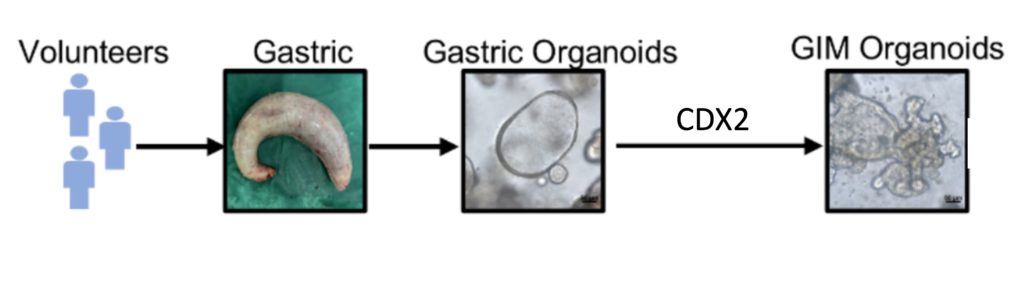
In examining which genes were turned up or down in the GIM organoids, the researchers found alterations in energy production. Since NAD+ plays an essential role in energy production, the researchers measured NAD+ levels. They observed low NAD+ levels in GIM organoids, four different metaplasia mouse models, and even human GIM tissue. Therefore, it would seem that low NAD+ levels contribute to GIM pathology.
Studies suggest that NAD+ declines with age, primarily due to an enzyme found on the surface of immune cells. The enzyme, CD38, consumes NAD+ by cleaving it into smaller molecules. Consistent with NAD+, the researchers found high CD38 levels in GIM organoids, four metaplasia mouse models, and human GIM tissue. These findings suggest that, in GIM, higher CD38 levels may lower NAD+ levels.
NAD+ Maintains Cellular Identity
NAD+ not only mediates the production of energy but also provides fuel for crucial enzymes. Notably, enzymes called sirtuins must consume NAD+ to function. As master gene regulators, sirtuins turn genes on or off. The researchers found that NAD+ depletion reduced sirtuin function, which turned off a gene that maintains the identity of stomach cells, called SOX2.
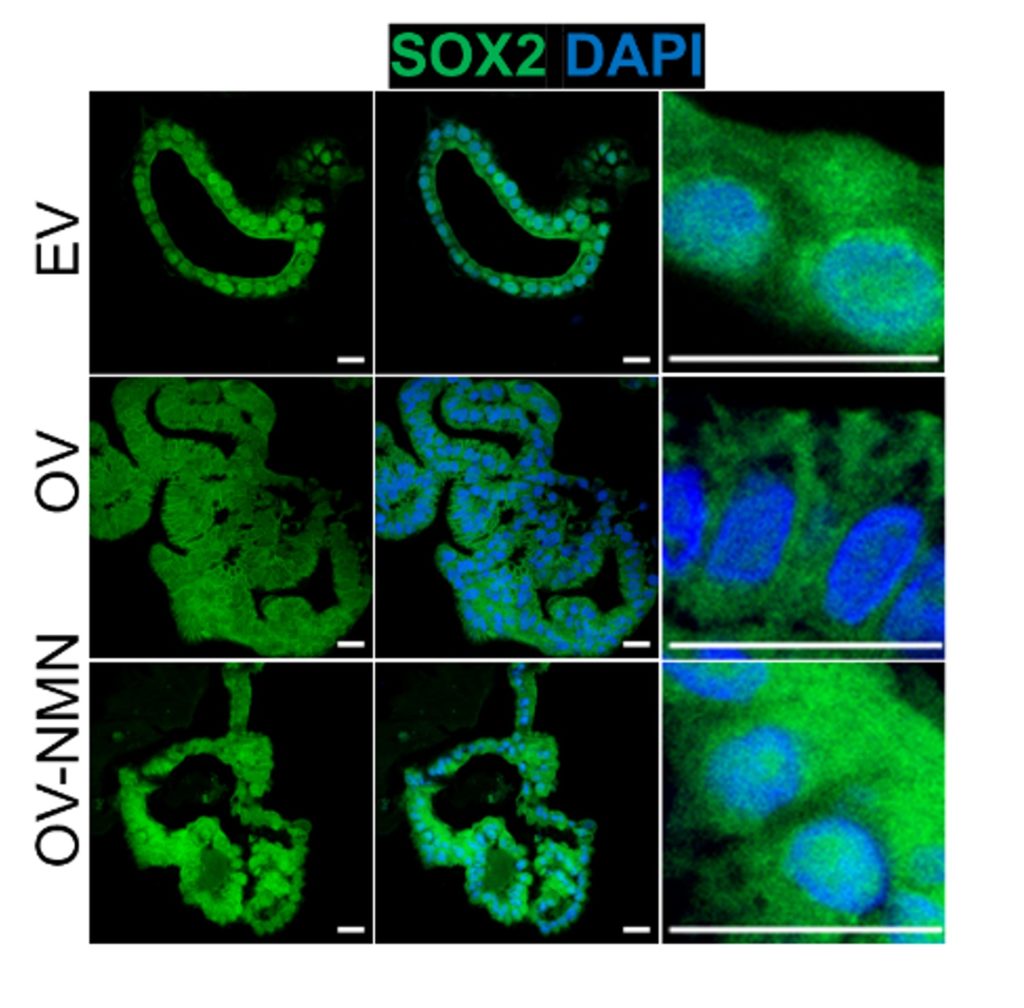
To determine if replenishing NAD+ could turn SOX2 back on, the researchers chose NMN. NMN, an NAD+ precursor, elevates NAD+ levels within cells. As suspected, they found that NMN turned SOX2 back on. Moreover, NMN relocated SOX2 to the nucleus of cells, where it has access to DNA to modulate genes. These findings suggest that NAD+ maintains the cellular identity of stomach cells by fueling sirtuins and keeping SOX2 activated.
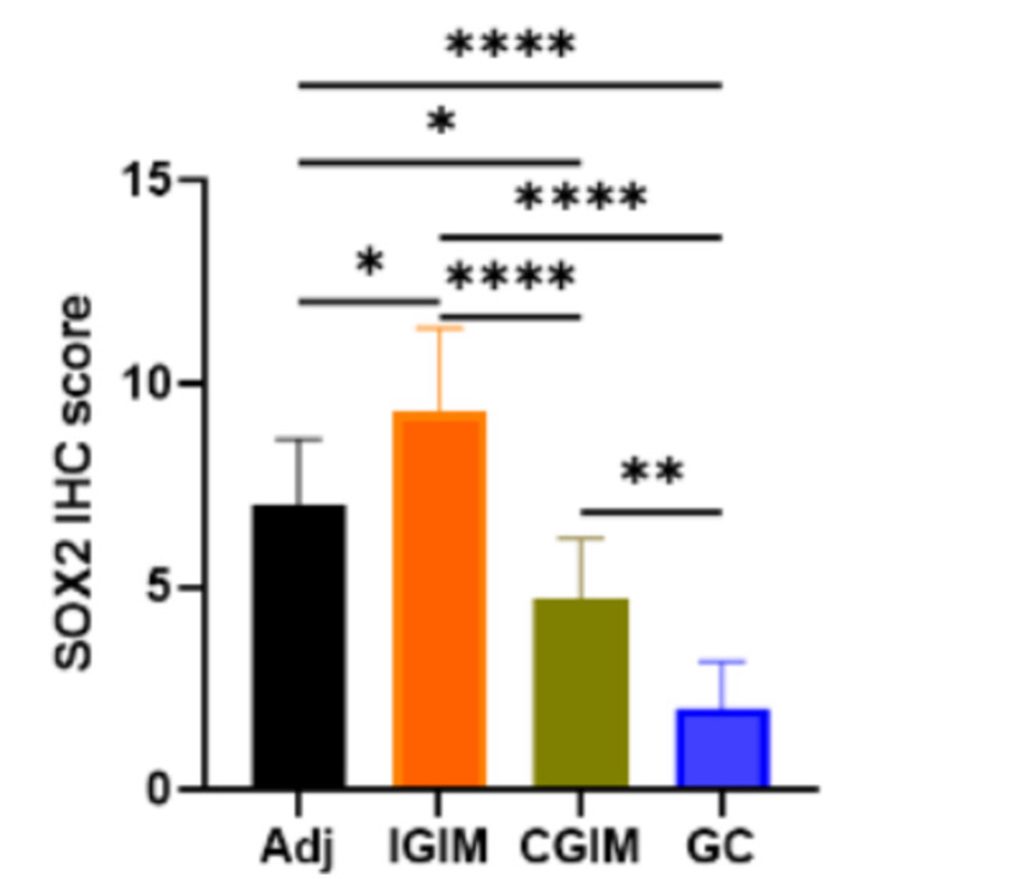
To ascertain whether SOX2 plays a role in human GIM, the researchers analyzed GIM tissue from donors. Using a technique called immunohistochemistry (IHC), they found that SOX2 was mostly located in the nucleus of normal stomach tissue. However, in GIM tissue, SOX2 levels were higher in the cytosol, where it is unable to function as a gene regulator. These findings suggest that SOX2’s localization to the cytosol may contribute to GIM pathology in humans.
NMN Reverses Metaplasia
To determine the effect of boosting NAD+ on the cellular mosaic that is GIM, the researchers applied NMN to GIM organoids. Strikingly, NMN shifted the cellular composition of GIM organoids to that of stomach organoids. NMN also lowered crucial proteins, such as the master gene regulator that triggered GIM, CDX2. Additionally, NMN elevated SOX2 in the nucleus, demonstrating its effects on maintaining stomach cell identity.
Furthermore, the researchers showed that NMN counteracts aspects of stomach aging and metaplasia in four different mouse models. For example, in a mouse model that lacks the gene responsible for producing stomach acid, NMN mitigated stomach lining deterioration. In mice infected by Helicobacter pylori, the most common cause of GIM, NMN reduced inflammation. Together, these findings suggest that NMN can reverse aspects of GIM.
Cellular Identity Loss Underlies All Cancers
Studies show that cellular identity loss, such as a stomach cell becoming an intestinal cell, underlies all cancers. Scientists call these changes in cellular identity cellular reprogramming, which is often modulated by NAD+-dependent sirtuin enzymes. Indeed, the latest research demonstrates that sirtuins play a large role in the suppression or progression of cancer.
Clinical researchers have already begun testing the effects of sirtuin modulators on cancer patients. For example, a 2010 study showed that the sirtuin activator resveratrol reduced tumor cell proliferation by 5% in colon cancer patients. In a 2013 study, the sirtuin inhibitor nicotinamide, combined with a cancer drug called vorinostat, stabilized the progression of lymphoma in patients who did not respond to other treatments.
Since sirtuins rely on NAD+ as fuel, it’s possible that in certain precancerous conditions, NAD+ boosters like NMN could be beneficial. Namely, considering that NMN may mitigate GIM, it could also mitigate the initiation of other precancerous tissues. However, once cancer is present, caution should be taken when consuming NAD+ boosters. Still, the Zhengzhou University study suggests that maintaining adequate NAD+ levels could play a preventative role in controlling the initiation of certain precancers.
Model: Four different mouse models for intestinal metaplasia
Dosage: 300 mg/kg/day of NMN administered via drinking water for 8 weeks

