NMN May Prevent Vascular Cognitive Impairment
Scientists from the University of Oklahoma Health Sciences Center conduct a study demonstrating nicotinamide mononucleotide (NMN) supplements improve brain blood vessel generation (crebromicrovascular angiogenesis) in aged rats.
Scientists from the University of Oklahoma Health Sciences Center conduct a study demonstrating nicotinamide mononucleotide (NMN) supplements improve brain blood vessel generation (crebromicrovascular angiogenesis) in aged rats. The scientists also point to studies underway to determine whether long term treatment with another nicotinamide adenine dinucleotide (NAD+) precursor, nicotinamide riboside (NR), improves blood flow to the brain in elderly adults with mild cognitive impairment. The scientists say, “If these studies yield positive results, the effects of NMN treatment on organ capillarization in elderly patients should also be investigated.”2
Age-associated functional and structural impairments of brain blood vessels (cerebral microcirculatory network) play a crucial role in disease development (pathogenesis) in the brain along with cognitive decline from aging.1,2,3,6,8,9,10,12 The brain uses the most energy of any organ.2 Nutrients and oxygen must travel through over 370 miles (over 600 kilometers) of blood vessels (cerebral microvessels) to reach neurons for proper brain function.2 Nearly each neuron has a blood supply from a microvessel (capillary).2 Recent research from the same group of scientists performing this study demonstrates treatment of older mice with NAD+ biosynthesis precursor, NMN, improves health of brain blood circulation (cerebral circulation) and improves blood supply to the brain.2,4 The present study seeks to find how NMN treatment affects age-related impairment of cells in the generation of brain blood vessels (endothelial angiogenic processes). Using brain blood vessel cells (cerebromicrovascular endothelial cells) from young and aged rats, the scientists test the proposal (hypothesis) long-term NMN treatment of the aged cells improves capacity to regenerate brain blood vessels. The study measures proliferation and migration of the brain blood vessel cells, along with their ability to form cellular structures resembling capillaries.
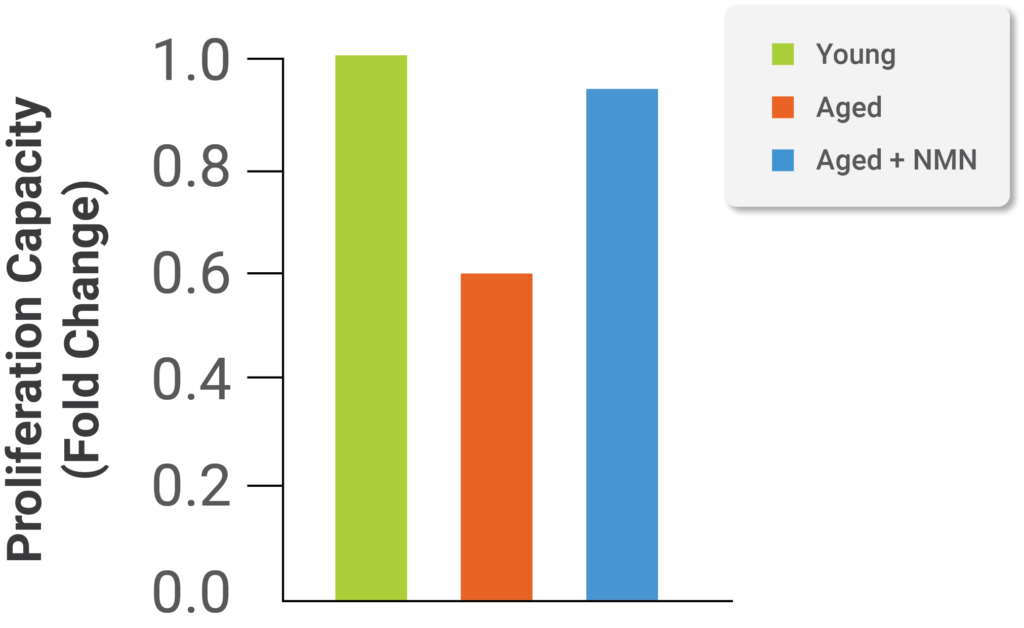
Image from Kiss et al. (2019)
Results from the study demonstrate proliferation of brain blood vessel cells decreases with age; aged cells have lower proliferation in comparison to young cells. Impaired proliferation in aged cells improves significantly with NMN treatment (see image above). In order to induce proliferation, scientists treated all three groups of cells with VEGF, a signal protein inducing generation of blood vessels (angiogenesis).
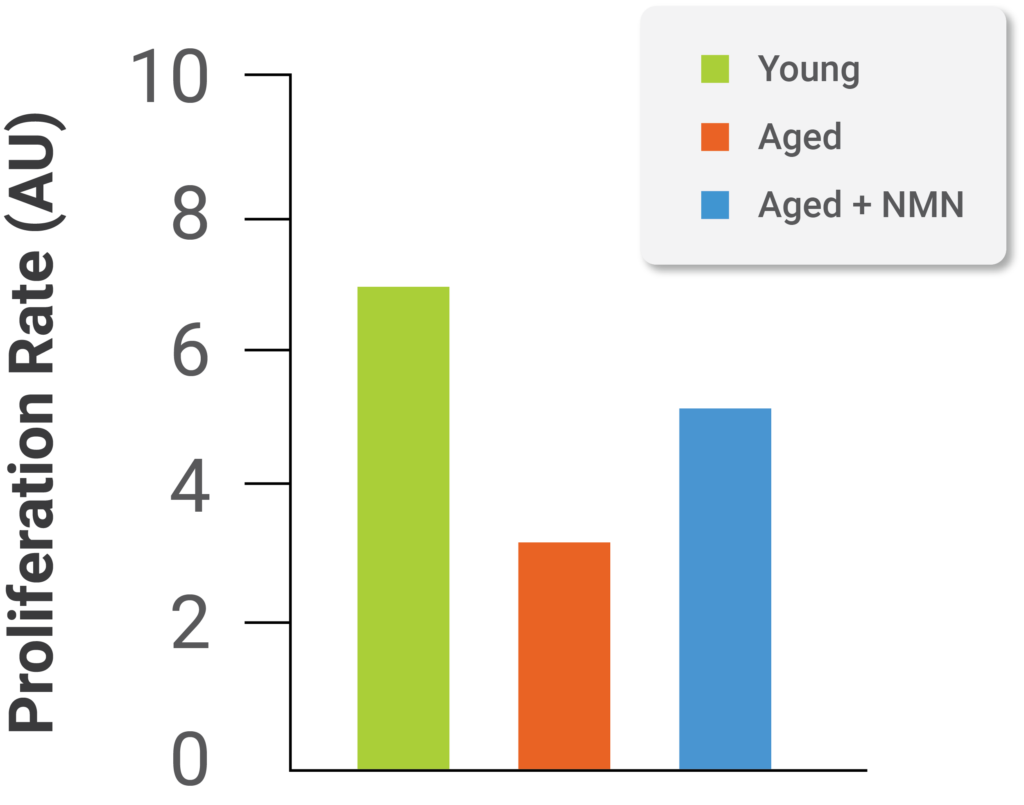
Image from Kiss et al. (2019)
Results also indicate migration of cells decreases significantly with age, and treatment of aged brain blood vessel cells with NMN improves migration of the cells almost to levels seen in young cells (see image above). The ability of brain blood vessel cells to migrate plays an essential role in blood vessel generation and blood vessel integrity.
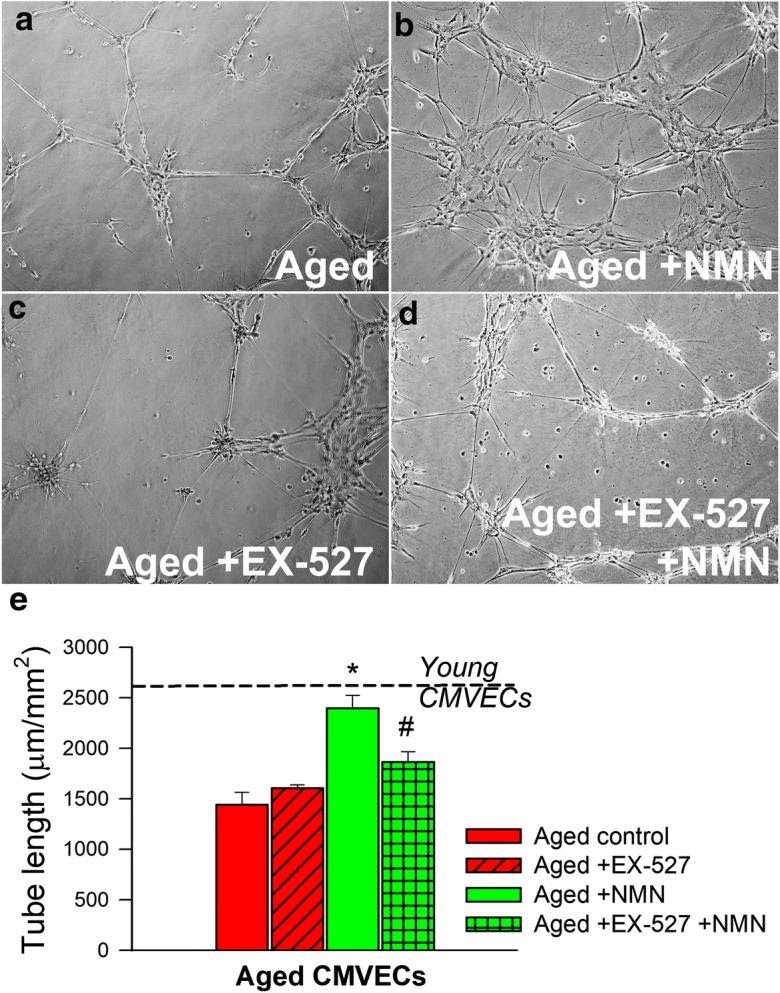
Image from Kiss et al. (2019)
The scientists demonstrate NMN treatment restores the ability of the aged brain blood vessel cells to form capillary-like connections (see ‘a’ and ‘b’ in image above). In images ‘c’ and ‘d’ of the image above, a pharmacological agent, EX-527, inhibits sirtuin activity. Sirtuins are proteins regulating cellular health. With pharmacological inhibition of sirtuins, the capillary-like connections between cells diminishes. Even with the addition of NMN treatment with EX-527 sirtuin inhibition, capillary-like structures between cells remains diminished. This provides evidence increased capillary-like connections between brain blood vessel cells with NMN treatment occurs through sirtuin activity. The graph in panel ‘e’ of the image above illustrates tube length of capillary-like structures from the cells increases in aged rat cells with NMN treatment.
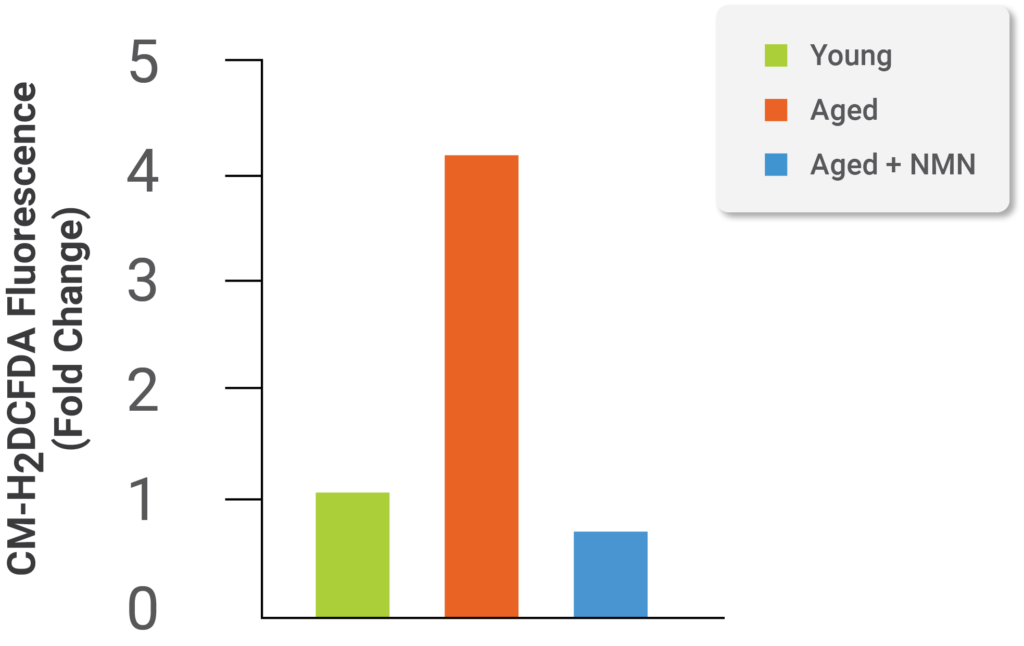
Image from Kiss et al. (2019)
Further experimentation in the study demonstrates NMN treatment of aged cells improves oxidative stress levels in aged cells, which resemble oxidative stress levels of young cells (see image above). According to the scientists of this study, “Our recent studies also demonstrate that attenuation of mitochondrial oxidative stress2,4,5,7,11 also restores endothelium-mediated vasodilation in aged mice.” The authors believe NMN and antioxidants targeting mitochondria, which reduce oxidative stress levels, improve blood flow to the brain in aging and have beneficial effects on brain function.2,4,5
NAD+ Precursors Promote New Blood Vessel Formation
According to the scientists, “The principal new findings of this study are that (1) age-related decline in cellular NAD+ levels is associated with impaired angiogenic response in aged rat CMVECs, and that (2) restoration of cellular NAD+ levels in aged CMVECs by treatment with NMN confers pro-angiogenic effects, counteracting, at least in part, the adverse effects of aging.” With age, reduced NAD+ levels in cells associate with impaired ability to generate new brain blood vessels. The study indicates restoring NAD+ levels in aged rat brain blood vessel cells (cerebromicrovascular endothelial cells) with NMN treatment provides benefits for generating new vessels, which could counteract effects of aging.

