New Study Shows Stem Cells Overcome Aging in Humans
Stem cell therapy improves mobility in older adults suffering from frailty—a condition characterized by increased vulnerability to disease, disability, and death.
Highlights
- Stem cells dose-dependently increase the distance individuals with frailty can walk in 6 minutes.
- A novel biomarker indicating the presence of functioning stem cells in the body was also identified.
Among older adults, frailty is a common condition defined as a state of heightened vulnerability to stressors, resulting in poor health outcomes. Older adults with frailty are susceptible to disability, mobility impairments, dementia, hospitalization, and death. However, frailty lacks an effective therapy, leaving those suffering to endure a reduced quality of life and impairments in daily living tasks.
To address this, researchers from Longeveron Inc. and the University of Miami Miller School of Medicine in Florida turned to stem cells. As published in their new study in Cell Stem Cell, the researchers are among the first to show that stem cell therapy counteracts aging, specifically frailty. These findings open the door for testing stem cells against other age-related conditions, like Alzheimer’s dementia.
Stem Cell Therapy Counters Age-Related Mobility Impairments
Mesenchymal stem cells (MSCs) are multipotent stem cells capable of differentiating into multiple cell types, including fat cells, cartilage cells, and bone cells. Due to their ability to repair tissue, modulate inflammation, and secrete bioactive molecules that support organ regeneration, they are the most commonly tested and clinically applied type of stem cells.
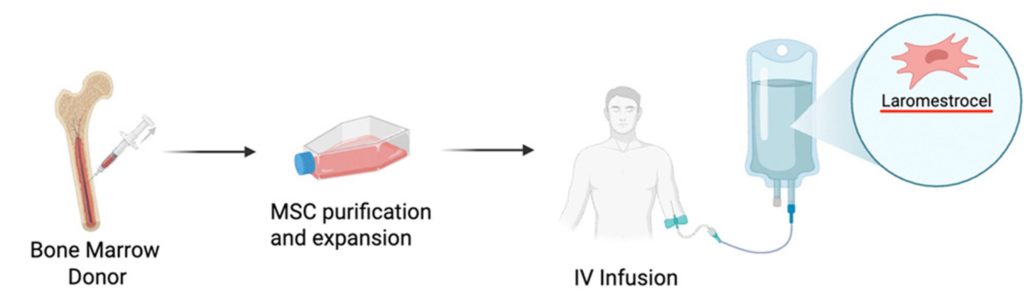
The Miami-based researchers took MSCs from the bone marrow of young adult donors, which were then purified, expanded, and stored. The participants of the randomized, placebo-controlled trial were then intravenously (IV) injected with either 25 million, 50 million, 100 million, or 200 million MSCs, which the researchers are calling Laromestrocel.
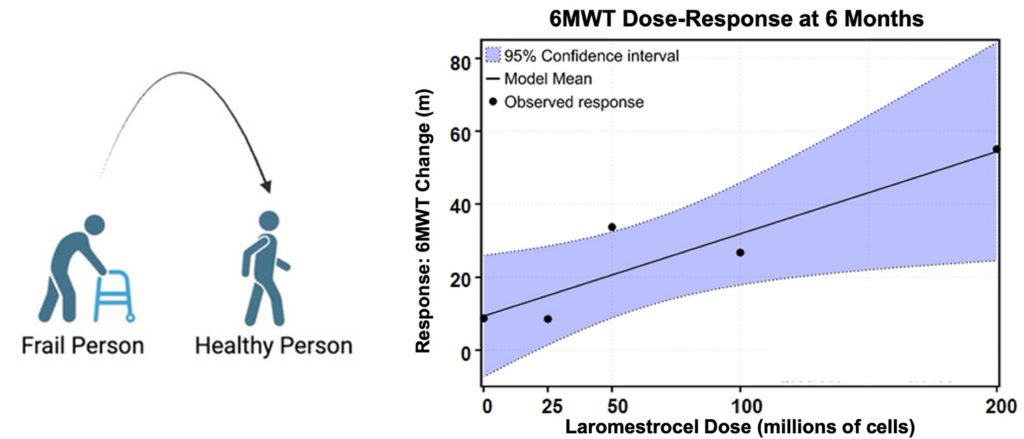
To assess the mobility impairments associated with frailty, the participants undertook a 6-minute walk test, where the distance walked in 6 minutes was measured. Remarkably, six months post-stem cell injection, the participants saw significant improvement in walking distance, which increased with higher doses of MSCs. These findings suggest that stem cell therapy can successfully treat the impaired mobility aspect of frailty and aging.
In addition to the 6-minute walking test, the participants filled out surveys scoring their subjective improvements in physical ability. The results showed no significant improvements in self-reported physical ability between the placebo group and the stem cell therapy groups. However, when including the placebo group, the improvements observed in the 6-minute walking test were correlated with subjective improvements in overall physical function, mobility, and upper extremity function.
A New Biomarker for Stem Cell Therapy
Biomarkers are measurable biological indicators that can be used to help scientists and physicians assess whether a treatment is having a physiological effect. As part of their study, the researchers sought to find a biomarker to assess the efficacy of their stem cell therapy.
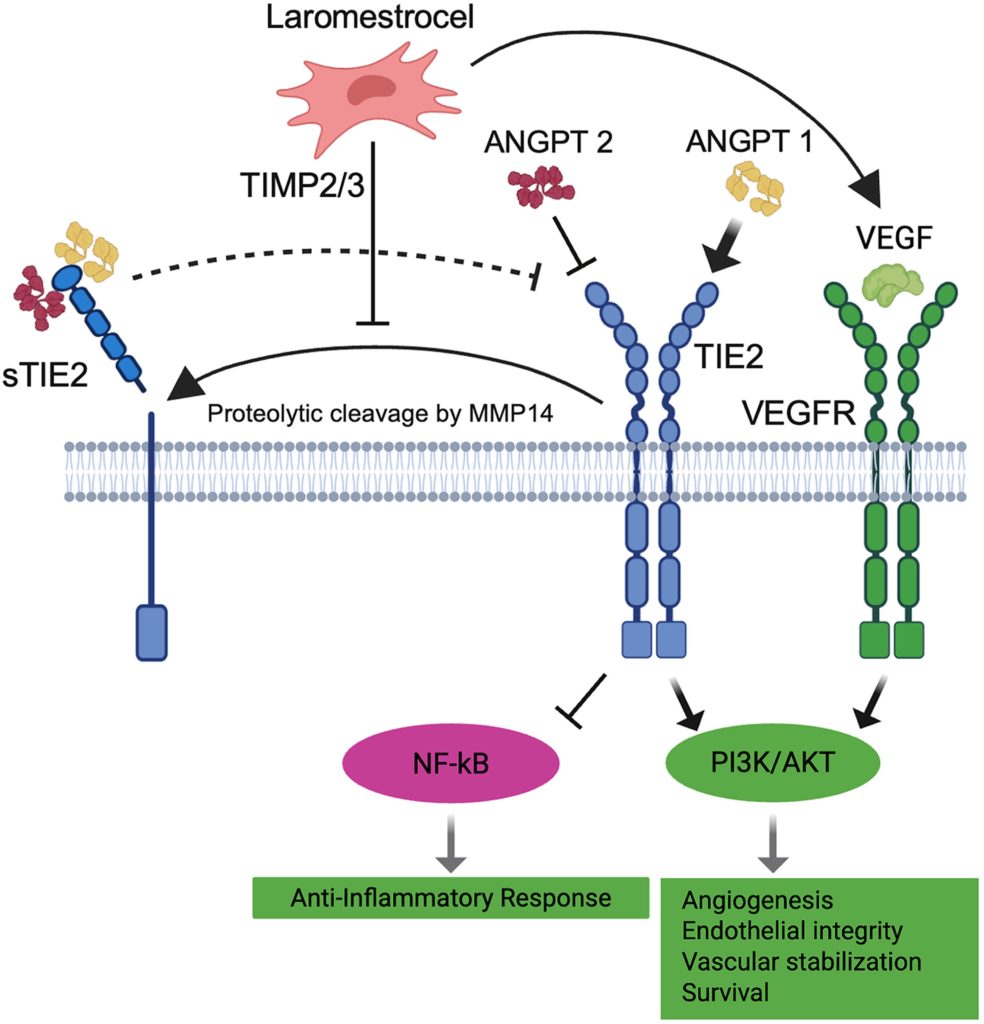
In screening 8 biomarkers, they identified a soluble protein called sTIE2 (soluble tyrosine kinase with immunoglobulin and epidermal growth factor homology domains) as the top candidate. TIE2 is a receptor that can be found within the inner lining of blood vessels (endothelial cells), where it improves vascular health and minimizes inflammation. However, under conditions of disease, including heart disease and psychiatric illness, TIE2 is cleaved, becoming sTIE2, which circulates through the bloodstream.
The cleaving of TIE2 into sTIE2 can be inhibited by certain molecules secreted by MSCs. Strikingly, the researchers found that sTIE2 was significantly reduced in response to 100 million stem cells. Moreover, a dose-response curve showed that sTIE2 levels remain low with stem cell numbers above 100 million. These findings point to the following model: MSCs block the cleavage of TIE2, allowing for decreased inflammation and improved vascular function, while lowering circulating sTIE2 levels.
Are Stem Cells the Future of Longevity Medicine?
Based on clinical studies, MSCs have a favorable safety profile. For example, a meta-analysis of 55 studies showed that MSCs increase the risk of fever, but not infection, cancer, death, or other adverse events. What’s more, previous studies have shown that MSCs improve physical function in frail individuals, as well as increase brain volume, reduce inflammation, and enhance cognition in Alzheimer’s patients.
These studies suggest that MSCs are capable of alleviating multiple aspects of aging, including physical and mental decline. Determining if MSCs can counteract other aspects of aging, such as heart disease and cancer, may just be a matter of time. This will depend on whether enough resources are available to interested scientists willing to conduct the clinical trials necessary to test MSCs against aspects of aging.
In December of 2024, the first MSC therapy was approved by the FDA (Federal Drug Administration) for the treatment of a rare condition called steroid-refractory acute graft versus host disease. This approval may open the door for the FDA’s approval of MSC therapies targeted against specific conditions of aging. Moreover, recent changes to the leadership of the United States Department of Health and Human Services may potentially fast-track such approvals.

