Can Pre-Workout NR Alter Human Metabolic Response to Endurance Exercise?
One week of nicotinamide riboside (NR) supplementation did not affect whole-body metabolic adaptation before, during, or after working out
Highlights
- One week of nicotinamide riboside (NR) supplementation doesn’t enhance human skeletal muscle metabolic adaptation before, during, or after endurance exercise.
- Signaling pathways sensitive to NAD+ in human skeletal muscle are not affected by this acute NR treatment either while working out for one hour, at rest, or in recovery.
The way our bodies turn food into energy, or metabolize, depends on how much we eat (or don’t) and how much physical (and mental) activity we exert. Our ability to adapt metabolically to these different situations allows our body and mind to get the most energetic bang for nutritional buck.
And at the center of metabolic adaptation is the molecule fundamental to cell function and survival called nicotinamide adenine dinucleotide (NAD+). Because our vitality and ability to adapt metabolically is tied to NAD+, there’s been a push to find effective NAD+ precursors that boost NAD+ levels. Although one precursor nicotinamide riboside (NR) has emerged as a dietary strategy to elevate NAD+ in animals, whether NR can elicit a similar response in humans has been unclear.
Now, researchers from the University of Birmingham in the UK and the Garvan Institute of Medical Research in Australia showed that NR supplementation (1000 mg/day) for one week did not alter human whole-body metabolism before, during, or after a one-hour aerobic workout. This acute NR supplementation neither augmented human skeletal muscle signaling implicated in metabolic adaptation to endurance exercise — in this case, using a cycling ergometer or stationary bike — nor affected the function and levels of mitochondria, the cell structures in charge of metabolism.
“Our data, therefore, adds to a growing list of studies suggesting that NR supplementation does not alter mitochondrial biogenic signaling in healthy human skeletal muscle,” concluded the authors in their article.
Nicotinamide riboside affects metabolism in rodents
NAD+ is considered indispensable for pathways governing our metabolic adaptations — how the body alters its efficiency at turning the food you eat into energy. In rodents, these processes can be modulated with NAD+ precursors. For example, oral NR supplementation promotes metabolic adaptation and induces cells to produce mitochondria in skeletal muscle. This is thought to occur, at least in part, by increasing the activity of longevity-linked proteins called sirtuins, which require NAD+ binding to function, to induce the production of mitochondria.
The effects of NR in humans is limited
But studies investigating NR supplementation in humans are in their infancy. Importantly, in humans, NR displays excellent safety and oral bioavailability — the extent a substance or drug becomes completely absorbed by the body for use. And studies have reported that NR supplementation can improve blood pressure, liver health, and physical function in the elderly, although the latter is not a consistent finding.
Despite the promising evidence in animals, several studies have reported no effect of chronic NR supplementation on metabolism, body composition, heart function, and endurance. On top of that, the effect of NR on mitochondrial biogenic signaling in human skeletal muscle following exercise remains unstudied. For these reasons, Stocks and colleagues investigated the effects of oral NR supplementation on whole-body NAD+ utilization and skeletal muscle mitochondrial signaling related to metabolism at rest and following endurance exercise in humans.
Acute NR does not alter human skeletal muscle
To do so, the international research team collected muscle biopsies before supplementation and pre-, immediately post-, and three-hours post-exercise (one-hour of cycling) performed following the supplementation period. The researchers postulated that NR supplementation would increase whole-body fat oxidation during exercise and augment SIRT1 and SIRT3 signaling in the post-exercise period compared to placebo. Contrary to their expectations, Stocks and colleagues found no such thing.
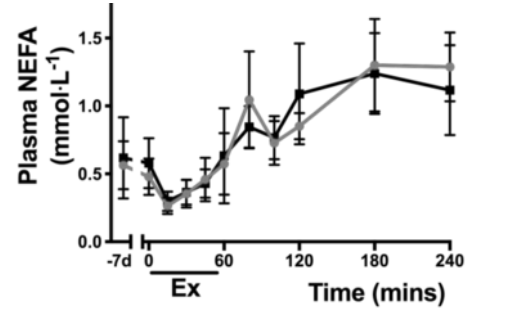
One week of twice-daily 500 mg NR supplementation in eight healthy human patients did not alter whole-body metabolism during and after exercise. Along these lines, there were no changes in the levels of key metabolic compounds — like glucose, lactate, and non-esterified fatty acids (NEFA) — during or after one hour of riding a stationary bike. Also, this acute NR treatment didn’t affect skeletal muscle mitochondrial function or production. So, it is not surprising that the researchers also did not observe the induction of signaling related to mitochondrial adaptation and production in resting or exercised human skeletal muscle of healthy human volunteers.
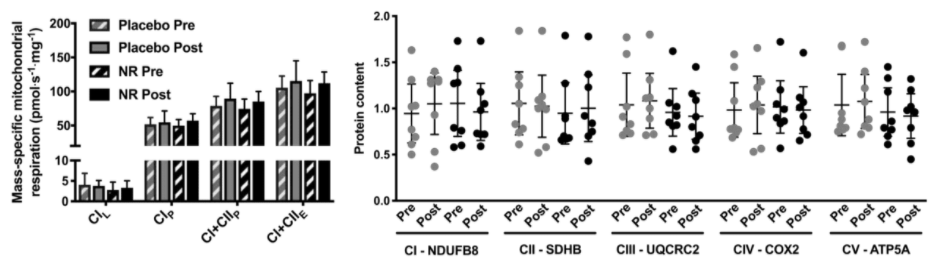
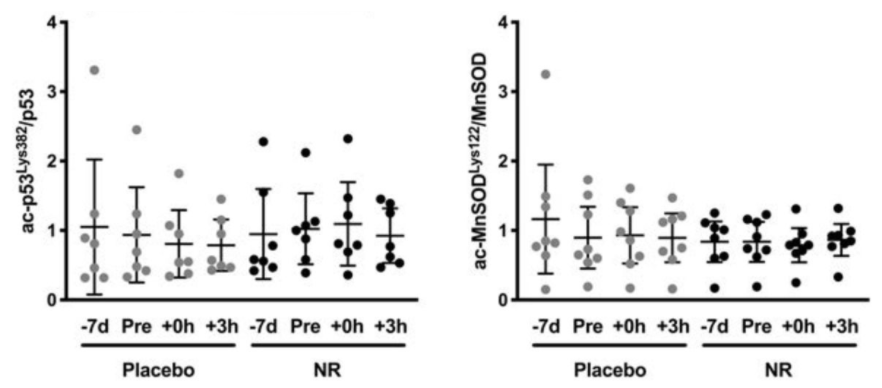
When Stocks and colleagues looked at whether there was any effect of NR supplementation on the activity of enzymes dependent on NAD+ to function, they found no changes compared to the skeletal muscle in untreated human participants. Contrary to the data showing that endurance exercise increases the activity of the sirtuins SIRT1 and SIRT3 in mice, NR did not appear to produce the same response in humans.
The researchers assessed this by analyzing the levels of modification in proteins regulated by SIRT1 and SIRT3. They found no change in these modification levels compared to untreated participants both at rest and in the post-exercise recovery period.
Although the researchers could show that the NAD+-precursor was bioavailable, NR supplementation did not increase skeletal muscle NAD+ concentration. Finally, and somewhat surprisingly, NR impaired the exercise-induced increase levels of NNMT — an enzyme proposed to play a generally reputed role in the whole-body metabolism of fat, which was contrary to their prediction.
What’s in store for future testing of NR in humans
“Collectively our data would therefore suggest that NAD+ metabolism is tightly regulated in human skeletal muscle, with short-term NAD+ precursor supplementation unable to modulate this response at rest or during and in recovery from endurance exercise,” said the authors. The chronic effects of NR on skeletal muscle NNMT content and activity, and whole-body and skeletal muscle fatty acid metabolism, warrant further investigation.

