Scientists Prolong the Life of Mice With Invisible Energy Fields, New Study Shows
Korean scientists use electromagnetic fields (EMFs) to trigger an anti-aging process known as cyclic cellular reprogramming in mice to extend their health and longevity.
Highlights
- Cellular reprogramming induced by EMFs increased the survival rates of aged mice.
- EMF-induced cellular reprogramming also rejuvenated aspects of the cardiovascular system, skin, liver, kidneys, and spleen.
- The researchers applied their EMF system to heal wounds, generate a superior mouse model for Alzheimer’s, and alleviate depression in mice.
Cellular reprogramming stands out as one of the most futuristic and borderline science fiction anti-aging interventions studied today. It involves reverting old cells into younger cells using only three genes. While gene therapy can be used to administer these genes, it can be invasive and fail to target specific tissues.
Now, scientists from Dongguk University in Korea have established a method for inducing cellular reprogramming with EMFs. As published in Cell, they show that EMFs prolong the lifespan of mice by turning on cellular reprogramming genes. They also show that this method counters aging in several organs. These findings suggest that cellular reprogramming therapy could become a reality in the not-too-distant future.
EMFs Prolong the Lifespan of Aged Mice
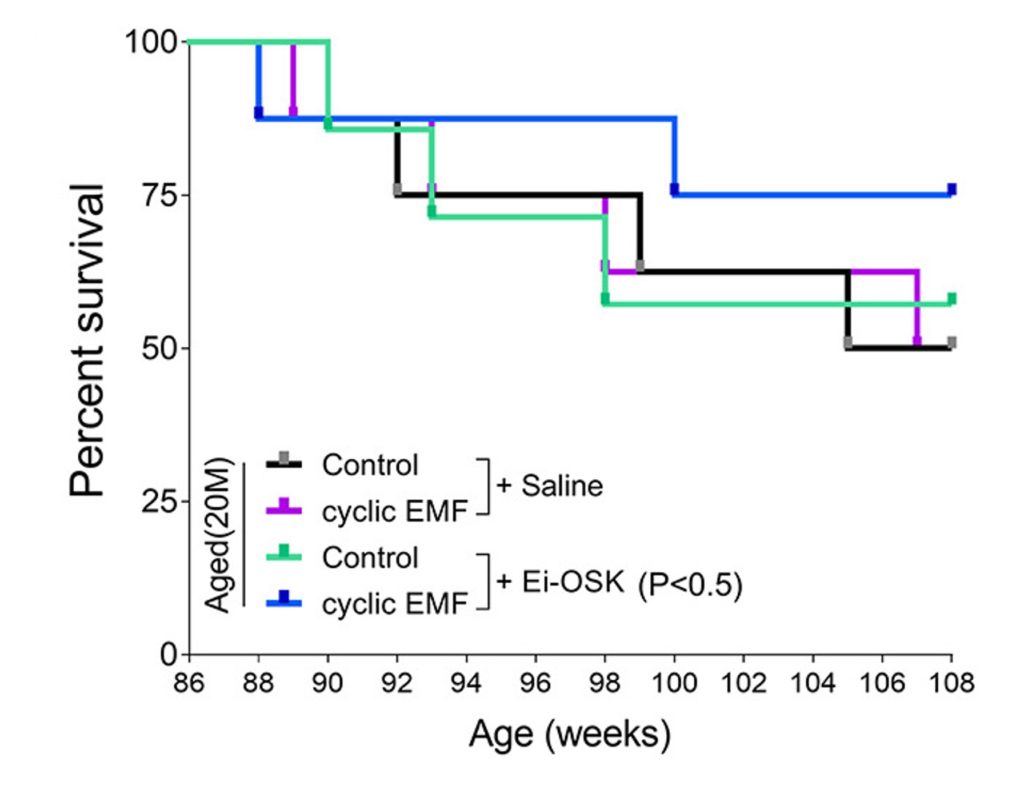
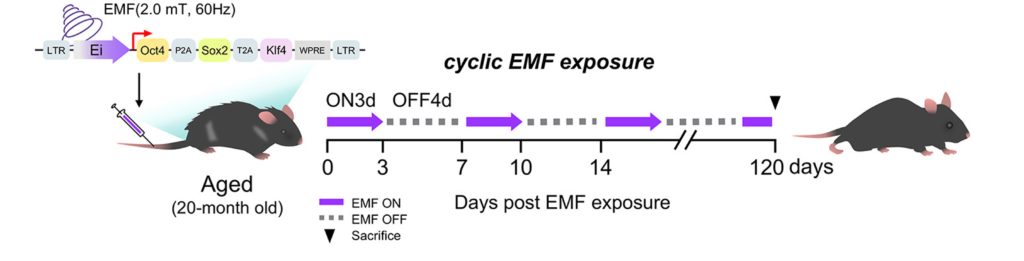
Complete cellular reprogramming can cause cancer and early mortality, so the researchers implemented cyclic cellular reprogramming. To do so, they genetically engineered aged mice to activate cellular reprogramming genes in response to EMFs. They applied the EMFs cyclically to induce cyclic cellular reprogramming. The researchers then assessed the survival of the mice up until they were 108-weeks-old, which is roughly equivalent to the human age of 70.
The researchers found that over 75% of the reprogrammed mice lived to 108-weeks-old. They also monitored the survival of engineered mice not exposed to EMFs. Only about 60% of these untreated mice survived until 108-weeks-old. To be thorough, the researchers also monitored normal-aged mice that were not genetically engineered. The survival rate for these mice was even lower, at about 50%. These findings suggest that EMF-induced cyclic cellular reprogramming can prolong the lifespan of aged mice.
EMFs Counteract Aging in Multiple Organs and Tissues
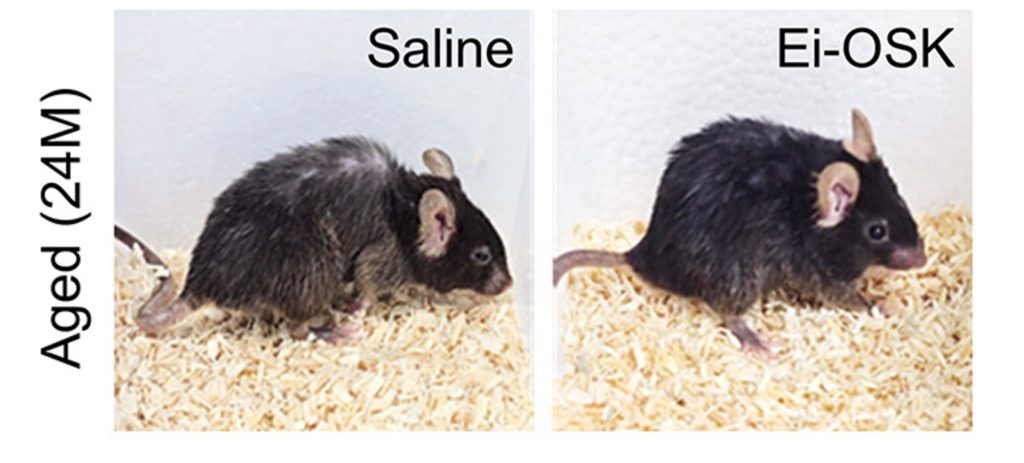
The Dongguk University researchers also found that EMF treatment countered certain aspects of aging in the engineered mice. The aorta, which thickens with age, was restored to normal thickness. Additionally, the treatment improved skin thickness and liver cell numbers, which decline with age, and it rejuvenated the spleen and kidneys. There were also signs of reduced senescent cells, which are cells that can accumulate with age and promote inflammation and tissue damage. The mice also become visibly younger, with less of a hunched back, better grooming, and a reduction in gray hair.
How Can EMFs Activate Genes?
The Dongguk University scientists developed a way to turn genes on and off using EMFs by utilizing a biological “switch.”
A Gene Naturally Activated By EMFs
They started by asking a simple question: which genes naturally respond to EMFs? In mouse brain tissue, they identified one gene in particular—called Lgr4—that could be activated and deactivated quickly. They then focused on the gene’s promoter, a stretch of DNA that modulates when a gene turns on or off. From this region, they chose a specific sequence and named it Ei, short for “EMF-inducible DNA element.”
A Natural EMF Sensor?
However, this did not explain how the EMFs actually trigger this switch. To find out, the researchers looked at what was happening inside cells. Their experiments suggested that EMFs interact with a protein called Cyb5b, setting off a chain reaction that releases calcium ions (Ca²⁺). Remarkably, the released Ca²⁺ oscillated at a frequency that activates the Ei switch.
Using EMFs to Activate Genes
With this mechanism in hand, the team engineered a controllable gene system. They used gene therapy to inject the cellular reprogramming genes—Oct4, Sox2, and Klf4 (collectively called OSK)—into older mice. These genes were placed under the control of the Ei switch, meaning they could be turned on or off simply by applying EMFs. In effect, EMFs became a remote control for gene activity.
The researchers also propose that Cyb5b, which was observed in human cells, may act as a previously unrecognized EMF sensor inside cells, a possibility that could open the door to new ways of interacting with biology using physical signals rather than drugs.
Additional Applications of the EMF System
On top of reversing aspects of aging, the researchers also experimented with additional applications for the EMF system:
- Wound Healing: To determine if Ei-OSK could be activated locally, the researchers injured the skin of mice. They then injected the Ei-OSK vector directly into the wound. Upon EMF exposure, this led to a reduction in scar (fibrotic) tissue, indicating improved wound healing.
- Alzheimer’s Disease (AD) Mouse Model: The researchers induced brain plaques in both aged and young mice using the EMF system. They found that aged mice exhibited accelerated plaque buildup, likely due to higher levels of brain inflammation. The memory of the aged mice was also impaired, demonstrating that the EMF system can be used to generate AD mouse models.
- Depression Alleviation: The researchers used the EMF system to reduce immobility, aggression, and anxiety-related measures in mice, suggesting the alleviation of depression-like behavior.
A Paradigm Shift in Partial Cellular Reprogramming?
Partial cellular reprogramming, where cellular reprogramming genes are not activated long enough (such as through cyclic activation) to completely revert cells into embryonic-like cells, has become a promising anti-aging intervention. The researchers say,
“The Ei gene switch offers a paradigm shift in in vivo partial reprogramming by addressing the critical safety concerns of current approaches and enabling its application in human therapeutics. Its rapid reversibility and non-invasive nature allow for precise control over OSKM expression, thereby preventing OSKM-driven hyperplasia.”
Hyperplasia, the rapid increase in cellular proliferation, can increase the risk of cancer, so preventing it is of utmost importance. This is why Life Biosciences, a company co-founded by Harvard’s David Sinclair, has removed one of the cellular reprogramming genes, c-MYC (the M in OSKM), like the Dongguk University researchers. Life Biosciences will be the first to test partial cellular reprogramming on humans. They will use gene therapy to deliver the OSK genes directly to the eye.
However, targeting deep tissues, particularly the brain, with gene therapy can be especially invasive. Targeting the brain with current gene therapy technology often requires drilling holes into the skull to inject the gene vector. Utilizing the EMF system can potentially be less invasive, as EMFs can target specific organs after the Ei-OSK gene vector has been injected through the skin. The researchers say,
“Moreover, by overcoming the inherent limitations of conventional transgenic systems in accessing human organs, this platform establishes a feasible framework for translating in vivo partial reprogramming into clinical applications for human age-reversal therapies.”

