Seragon Reports Record-Breaking Data on SRN-901, a New Drug That Extends Mouse Lifespan by 33%
In a late-life mouse study, the multi-component drug candidate increased remaining lifespan by 33%, slowed frailty progression, and reduced tumor incidence, according to data published in Drug Design, Development and Therapy.
Highlights
- SRN-901 increased median remaining lifespan in adult mice by 33% in a late-life intervention study.
- The treatment was associated with a 70% attenuation of frailty progression and a 30.53% reduction in tumor incidence.
- Gene expression and metabolite analyses suggested that SRN-901 influenced pathways linked to aging, inflammation, antioxidant defense, and metabolism.
Aging is a multifactorial process marked by declining physiological function, rising frailty, and greater vulnerability to age-related disease. To address that complexity, Seragon developed SRN-901 as a combination therapy designed to act on several pathways associated with aging at the same time. That strategy stands apart from the many longevity studies that focus on single agents.
Now, in a study published in the journal Drug Design, Development and Therapy, researchers at Seragon in Irvine, California, report that SRN-901 significantly extended remaining lifespan in adult mice by 33%. The treatment was also associated with a 70% attenuation of frailty progression and a 30.53% reduction in tumor incidence. Gene expression analyses suggested downregulation of pathways linked to age-related disease, including Alzheimer’s disease-related pathway annotations, while metabolite profiling pointed to changes in pathways associated with aging and longevity. Taken together, the findings support the idea that a multi-component intervention may influence several biological features of aging at once.
More Information on SRN-901
SRN-901 combines several compounds with roles in cellular maintenance and stress response that are intended to act across interconnected pathways tied to mitochondrial maintenance, inflammation, metabolism, and cellular resilience. That multi-pathway design reflects a broader view in aging biology: because aging affects many systems at once, interventions aimed at only one target may have limited effects.
SRN-901 Extended Lifespan and Improved Markers of Healthspan
To evaluate the effects of SRN-901 on late-life survival, the researchers studied 18-month-old C57BL/6 mice, which the article describes as roughly comparable to 56-year-old humans. The animals received SRN-901, rapamycin, nicotinamide mononucleotide (NMN), or NR, and were followed until humane end-of-life criteria were reached.
The SRN-901-treated mice showed a 33% increase in median remaining lifespan. Treatment was also associated with a 46% reduction in the hazard of death. Rapamycin significantly extended lifespan as well, but not to the same extent as SRN-901, while NMN and NR did not significantly improve lifespan in this experiment. Those results suggest that SRN-901 produced a comparatively strong survival signal in this late-life mouse model.
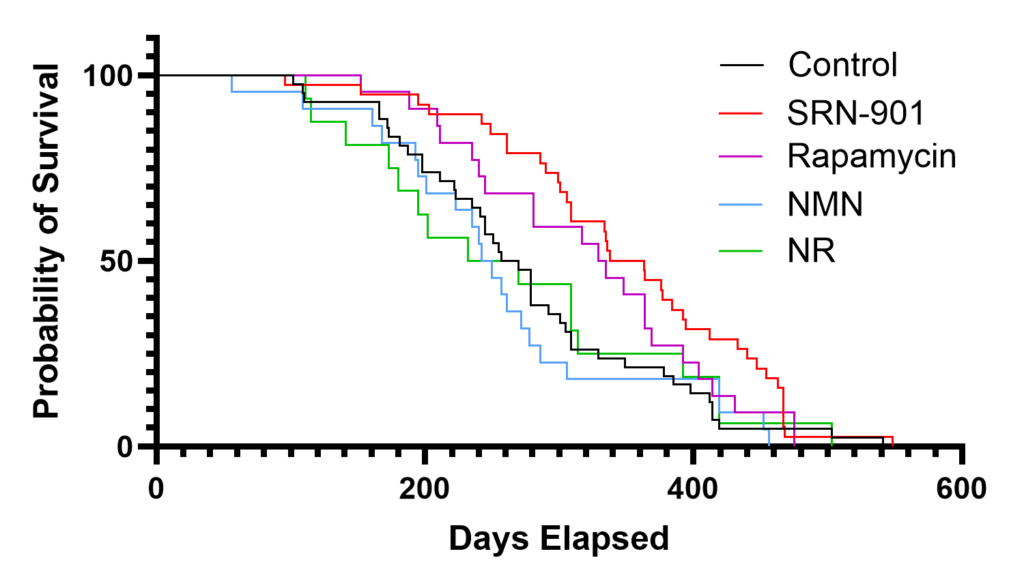
The researchers also assessed frailty, which is commonly used as an inverse marker of healthspan. Frailty testing performed before and after treatment indicated a 70% attenuation of frailty progression in the SRN-901 group. In practical terms, the data suggest that the added lifespan was accompanied by slower functional decline rather than simply more time lived in poor health.
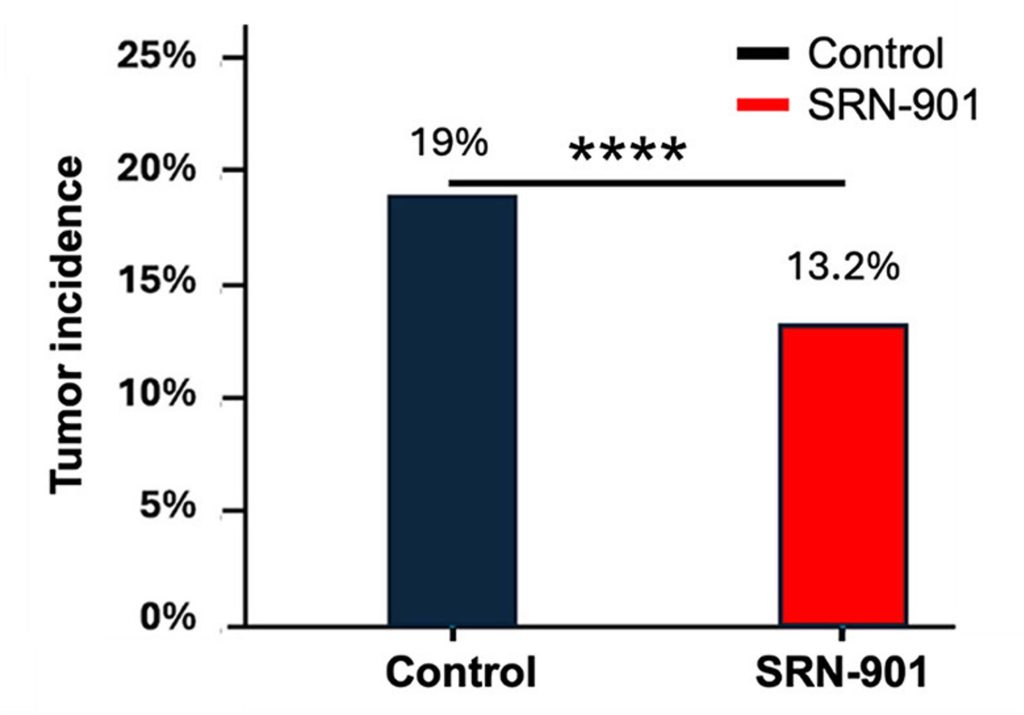
Tumor incidence was also lower in the treated animals. At the end of life, the researchers found a 30.53% reduction in tumor incidence in mice that received SRN-901, providing additional evidence that the treatment may have affected broader age-related decline.
What the Molecular Data Suggest
To better understand the biology behind those outcomes, the researchers analyzed gene expression in blood samples collected before treatment and again 56 days after treatment began. Their results suggested significant modulation of age-associated pathways. Among the most notable findings was downregulation of pathway annotations related to Alzheimer’s disease, which the authors interpret as evidence of a shift away from molecular signatures associated with aging and neurodegeneration.
The team also profiled metabolites in the blood to examine whether SRN-901 altered systemic metabolism. Those analyses suggested upregulation of pathways involving glutathione metabolism, insulin signaling, and FoxO signaling. Because those pathways are widely linked to antioxidant defense, metabolic regulation, and cellular repair, the findings offer a possible mechanistic explanation for how SRN-901 may have influenced both lifespan and frailty in the animals.
What Comes Next
The study adds to growing interest in combination approaches to longevity research, but it does not establish that SRN-901 will extend lifespan in humans. Determining whether similar effects occur in people would require clinical testing, and studies built around lifespan itself would take many years to complete.
Researchers could instead look for earlier signals in human trials, such as changes in biological aging markers over six months to a year. One example is epigenetic age, which estimates biological aging by measuring chemical modifications to DNA. Some research suggests that accelerated epigenetic aging is associated with higher mortality risk. If future trials show that SRN-901 favorably affects such biomarkers, that could offer an earlier indication of whether the drug influences human aging biology.
For now, the mouse data position SRN-901 as a notable preclinical candidate in a field that is increasingly exploring whether multi-target interventions can outperform single-agent approaches.
“Our findings demonstrate that SRN-901 administration significantly increases the lifespan of mice, reduces frailty scores, modulates proteomic and gene expression pathways linked to longevity and aging-related diseases, and inhibits metabolic aging,” the Seragon researchers wrote.
| Model | C57BL/6 mice |
| SRN-901 | 500 mg/kg/day via oral gavage, six days per week, beginning at 18 months of age |
| Rapamycin | 14.4 ppm mixed in food, beginning at 18 months of age |
| NMN | 300 mg/kg/day via oral gavage, six days per week, beginning at 18 months of age |
| NR | 300 mg/kg/day via oral gavage, six days per week, beginning at 18 months of age |

