Silver Quantum Dots Enhance Liver Absorption and Therapeutic Effectiveness of NMN and Metformin
NMN and metformin bound to microscopic nanocrystals called silver quantum dots enhance metabolic function using substantially lower doses in mice.
Highlights
- Attaching NMN and metformin to silver nanocrystals called quantum dots significantly enhances their uptake into liver cells.
- The quantum dot-bound NMN and metformin facilitate improved metabolic responses like blood glucose clearance at much lower drug doses.
Nanomedicine is the application of nanotechnology – technology dealing with tiny dimensions of less than 100 nanometers – to therapeutic agents. Some believe biotechnology research has reached “the beginning of the end of the nanomedicine hype” mostly due to the lack of nanotechnology’s translation to treat cancer. At the same time, since 80-95% of nanomaterials accumulate in and are cleared by the liver, others propose nanomedicines have considerable promise in targeting liver diseases and metabolic disorders. Whether nanotechnology application to therapeutics targeting liver disease and metabolic disorders enhances their effectiveness remains to be explored.
Cogger and colleagues from the University of Sydney in Australia published a study in ACS Nano demonstrating that attaching nicotinamide mononucleotide (NMN) and metformin to silver nanocrystals called quantum dots substantially enhances their uptake in the liver. The quantum dot-bound medicines also facilitate an improved metabolic response to the sugar glucose, indicative of enhanced metabolism. Enhanced drug delivery and potency with quantum dots may translate to improved drug delivery methods for aged individuals with metabolic or liver disease.
The Effects from NMN and Metformin Treatment
NMN is a precursor to the vital molecule nicotinamide adenine dinucleotide (NAD+) that has essential roles in cell energy-generating processes. By increasing NAD+ levels in mice, NMN can also drive the activation of proteins dependent on NAD+ called sirtuins that promote metabolism.
The other quantum dot-conjugated drug explored in the study, metformin, is extensively prescribed for type 2 diabetes. It increases lifespan in animal models by adding a molecular tag and thereby activating the enzyme referred to as adenosine monophosphate-activated protein kinase (AMPK). AMPK triggers the uptake of glucose when cellular energy is low thereby enhancing metabolism.
NMN’s stimulation of sirtuin function and metformin’s activation of AMPK can both promote improved metabolic function. To find out if binding NMN and metformin to quantum dots for oral drug delivery through water consumption improves their potency, Cogger and colleagues measured the cellular uptake of the quantum dot-bound pharmaceuticals along with the clearance of the sugar glucose in blood.
Quantum Dots Enhance Liver Cell Absorption of Metformin
The Australia-based research group first examined quantum dot-bound metformin uptake in the liver. They saw expedited absorption with higher cell metformin concentrations in liver cells following oral ingestion. While metformin alone peaked at eight hours after consumption, the quantum dot metformin levels peaked much more quickly at about two hours. Quantum dot binding to metformin also enhanced the total amount of metformin that the liver absorbed. These findings indicate that quantum dots substantially improve therapeutic agent absorption in the liver with faster uptake in greater quantities.
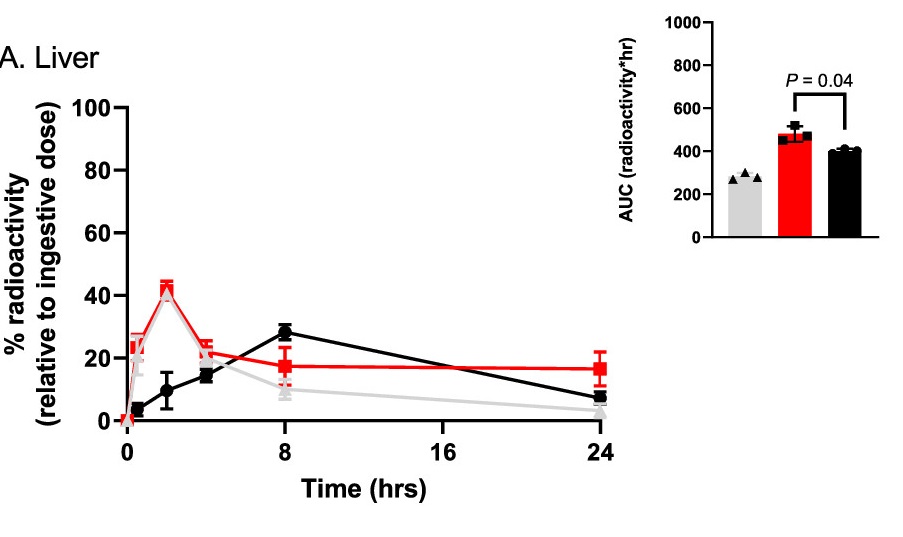
Less NMN and Metformin Are Needed with Quantum Dots to Clear Blood Glucose
Cogger and colleagues then found that the medicines conjugated to quantum dots provided significantly reduced blood glucose levels in tolerance tests for the sugar that measure the body’s ability to clear it. Glucose tolerance tests measure blood sugar concentrations after ingesting a glucose solution, and type 2 diabetes patients cannot typically clear blood glucose. To achieve the same glucose tolerance test results in mice, the metformin dose required with quantum dots was 100 times less than that of metformin alone. NMN, although not as potent at inducing glucose clearance, required a thousand times less of a dose compared to NMN alone. These findings indicate that quantum dots greatly enhance therapeutic agent absorption so that drastically diminished doses of the medicine are required with quantum dots.
Clathrin Proteins Mediate Cellular Quantum Dot Uptake
To see out how quantum dots improve uptake, Cogger and colleagues examined how cells absorbed these compounds. They found liver cells use a cellular uptake mechanism utilizing proteins that create small sacs to absorb the quantum dots called clathrin-mediated endocytosis. The medicines can bypass cellular transporters that typically absorb them and instead get absorbed by clathrin-mediated endocytosis. For example, the transporter OCT1 absorbs metformin on its own, but inhibiting OCT1 did not reduce the uptake of quantum dot-bound metformin. This shows that quantum dot conjugated medicines could have better uptake by getting around their typical absorption transporters, which can deteriorate with age. Bypassing deteriorating transporters for medicine absorption with quantum dot-bound medicines could provide a means to enhance medicine uptake in aged individuals.
“We show that [quantum dot]-metformin and [quantum dot]-NMN have dramatically increased hepatic bioavailability and 100- to 1000-fold improvements in metabolic effects compared to drug alone,” stated Cogger and colleagues in their publication.
Future Nanomedicine Research on Liver Dysfunction
Based on their findings, the research team says quantum dots are ideal for nanotherapeutic development. More work should be geared toward developing nanotherapeutics that target the liver, rather than other organs, since the liver absorbs and clears most nanomedical agents.

