The First Non-Invasive Indicator to Evaluate Drugs that Target Aging Cells
This indicator can facilitate research and clinical trials of drugs that target age-related diseases
Highlights
· Non-replicated cells linked to aging make several bioactive fat molecules, including a type called prostoglandins
· One specific prostoglandin called dihomo-15d-PGJ2 is released when these non-replicating cells are destroyed
The hunt for anti-aging drugs has been continuously gaining steam. One major approach to prevent aging has been to develop senolytics — drugs that target cells that have stopped growing and replicating, an age-related phenomenon called senescence that has been shown to limit lifespan. But there’s one major problem with senolytics, which is that there’s no real great way to evaluate how well they are working in an animal let alone people.
In an article published in Cell Metabolism, Wiley and colleagues from the Buck Institute for Research on Aging identify an indicator of senescence that can be analyzed in live subjects. They found that senescent cells make a fat molecule called 15d-PGJ2 that is released upon their death. These findings can be used towards evaluating the effectiveness of senolytics, several of which have already entered clinical trials.
Cell cycle arrest is linked to aging
Cellular senescence is a term that has become synonymous with aging. Senescent cells can drive several age-associated diseases in part by generating a myriad of biologically active molecules. These molecules, which include secreted inflammatory molecules that can have potent local — and potentially systemic — effects on tissues. We also know that senescent cells increase with age in human and mouse tissues, which, at least in mice, limits both median lifespan and healthspan. But all the components of these senescence-associated products have not all been looked at carefully.
Bioactive fat molecules are indicators of senescence
Here, Wiley and colleagues show that senescent cells also synthesize many biologically active fat molecules that can reinforce this proliferative arrest. One such class that they identified are known as prostaglandins — a group of fat molecules that control processes, such as inflammation, blood flow, the formation of blood clots, and the induction of labor.
Specifically, they found that one member of this family of fat molecules 15d-PGJ2 is both a biomarker and promoter of senescence. When they treated cells with 15d-PGJ2, the cells activated markers of senescence and stopped proliferating. What’s more, the research team from the Buck Institute for Research on Aging found that 15d-PGJ2 is a biomarker of senolysis — when senescent cells die and deteriorate, releasing their contents into the surroundings. In both cultured cells and in living animals, Wiley and colleagues found that 15d-PGJ2 spills out of the dying senescent cells.
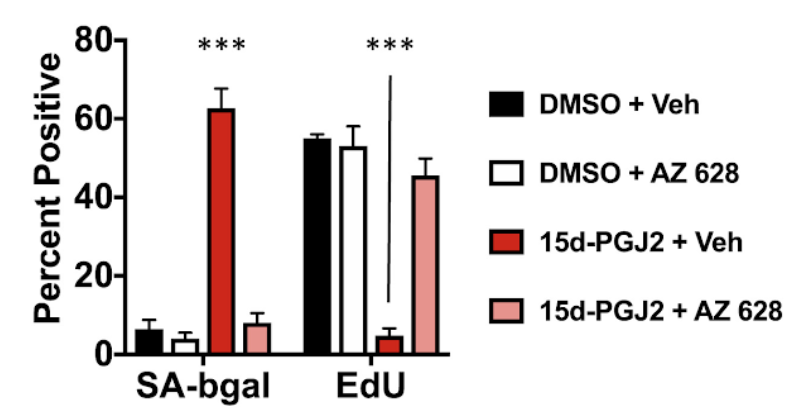
“Lipid components of [senescence] have been vastly understudied,” says lead scientist Christopher Wiley, Ph.D. “The biosynthesis of these signaling lipids promotes segments of the SASP and reinforces the permanent growth arrest of senescent cells. This work provides a new way of understanding and studying senescence-driven pathology,” he said.
15d-PGJ2 can be used to evaluate anti-aging drugs
Wiley and colleagues propose that using 15d-PGJ2 as a biomarker for senolysis has several potential applications. Senolytic drugs are increasingly being used in aging and related research and, importantly, have entered early clinical trials. Determining that senolysis is taking place is essential for evaluating these compounds as therapeutic agents. Detection of 15d-PGJ2 in biological fluids may allow rapid evaluation of the efficacy of these compounds.
“We hope that identifying and including these bioactive lipids as part of the SASP will encourage researchers working in a broad range of fields to take a new look at cellular senescence,” said Buck professor Judith Campisi, PhD, senior scientist on the study. “The fact that one of these lipids ends up being a simple non-invasive biomarker for tracking the efficacy of treatments is a huge plus for those of us working to stem the ravages of age-related disease.”
“The list of age-related diseases definitively linked to cellular senescence keeps growing, as does the number of biotech companies racing to develop drugs to eliminate senescent cells,” said Campisi. “While the field has never been more promising, the lack of a simple biomarker to measure and track efficacy of these treatments has been a hindrance to progress. We are excited to bring this new biomarker to the field and look forward to it being used in the clinic.”

