University of Pittsburgh Finds Stem Cell-Secreted Particles That Can Restore Cognitive Function in Old Age
Researchers find that injecting aged mice with particles secreted from stem cells (extracellular vesicles) enhances memory.
Highlights:
- Injecting aged mice with young mouse serum containing extracellular vesicles ameliorates memory impairment.
- Removing EVs from young mouse serum reverses the serum’s cognitive-boosting effects.
- Treatment with EVs increases the activity of several genes involved in neuroprotection.
Extracellular vesicles (EVs) have gained prominence in the field of neuroprotection due to their emerging roles as critical intercellular communicators within the nervous system. These tiny, membrane-enclosed structures, originating from diverse neural cell types, carry a cargo of essential bioactive molecules that hold promise for safeguarding neurons. With this in mind, researchers continue to investigate the intricate molecular mechanisms of EV-mediated neuroprotection to pinpoint potential avenues for therapeutic advancements in neurodegenerative disorders and brain injuries.
Now, in a new study reported in the International Journal of Molecular Sciences, researchers from the University of Pittsburgh explored whether injecting aged (22-24 months old) mice with young mouse serum containing EVs (YS) can ameliorate age-related cognitive dysfunction. Behavioral memory tests revealed that YS injections successfully improved cognition. Furthermore, Fitz and colleagues demonstrated that YS injections increased the activity of multiple genes involved in neuroprotection; however, the neuroprotective effects of YS injections were reversed when the investigators removed the EVs from the young mouse serum, highlighting their importance.
Young Serum EVs Protect Against Cognitive Decline
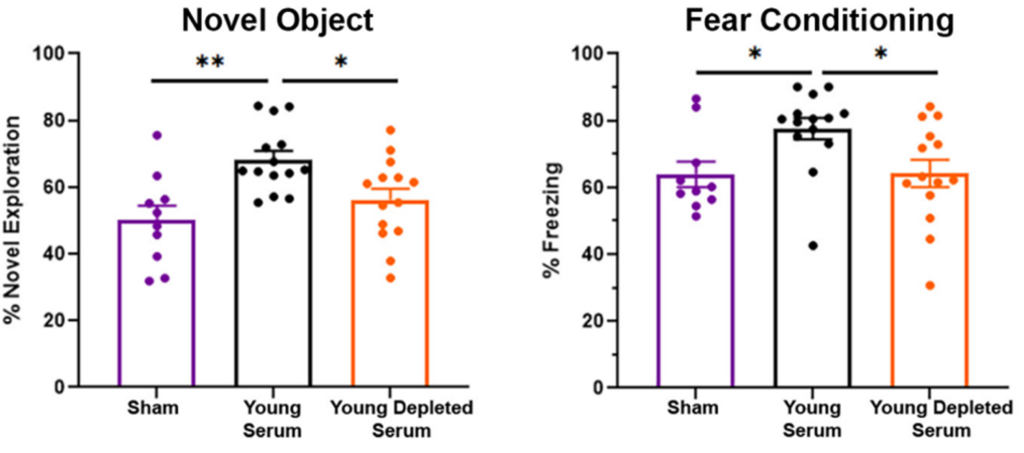
Along with EVs from young serum carrying tissue-preserving molecules, their ability to readily enter the brain highlights their potential to restore an aging brain to a youthful state where cognition is functional. To explore this, Fitz and colleagues injected naturally aged mice with YS and assessed its effects on learning and memory. Additionally, the investigators injected one group of mice with young serum depleted of EVs to further elucidate the role of EVs in preserving cognition.
The investigators subjected mice to two common memory tests – novel object recognition test; contextual and cued feared memory test – and found that YS injections significantly boosted cognitive performance, with treated mice exhibiting superior learning and memory than untreated mice. However, the investigators noted that depleting young serum of EVs reversed the treatment’s neuroprotective effects, demonstrating that the cargo inside EVs is necessary to positively affect brain function.
Young Serum EVs Activate Brain-Boosting Genes
While Fitz and colleagues established the importance of EVs in enhancing brain function, they weren’t able to determine the specific factors in the EVs’ cargo responsible for their brain-boosting effects. That being said, the investigators were intrigued about EVs’ effects on brain gene activity, specifically those involved in neuroprotection.
The investigators first looked at the activity of genes involved in the integrity of the blood-brain barrier (BBB), which facilitates the passage of ions, nutrients, and other molecules to maintain a stable and optimal chemical environment for neurons to function properly. Upon analysis, the data revealed that YS increased the activity of several genes involved in stabilizing the BBB, highlighting a potential mechanism behind EVs neuroprotective effects.
The Pittsburgh researchers proceeded to analyze the activity of genes involved in the health of neurons, which are critical for the transmission, processing, and integration of information. The results showed that YS treatment increased the activity of genes that stimulate the formation of synapses – neuronal junctions that allow electrical or chemical signals to be transmitted between neurons. Furthermore, YS treatment increased the activity of genes involved in the growth of axons – long, slender projections of neurons that transmit electrical impulses away from the cell body to communicate with other neurons, muscles, or glands. Collectively, the findings suggest that YS exerts neuroprotection by boosting neuron-preserving genes.
Harnessing EVs to Prolong Longevity
In addition to EVs exerting neuroprotective effects, previous studies have shown that injecting aged mice with EVs also counters age-related physical decline and drives muscle tissue regeneration. Furthermore, EVs have been shown to thwart one of the primary hallmarks of aging: senescent cells. These dormant cells sprout across the body’s organs as we age, driving tissue deterioration and the progression of age-related diseases. Thus, EVs’ ability to selectively target and eliminate senescent cells opens up a new avenue for delaying multiple features of aging.
Notably, a recent study found that EVs successfully increased the lifespan of old rats while simultaneously improving heart function. So, while it’s clear that EVs show promise in promoting longevity, additional research is required to pinpoint the exact molecules present in EVs’ cargo responsible for their health-boosting effects.
Model: 22-24 month old male wildtype C57/BL6 mice; Young male wildtype C57/BL6 mice
Dosage: 100 uL of young serum containing extracellular vesicles (EVs)

