NMN and 78c: A New Synergy for Treating Dry Eye and Aging
NMN (nicotinamide mononucleotide) enhances the effect of 78c, which inhibits an NMN-degrading enzyme called CD38, against age-related dry eye in mice.
Highlights
- The NAD+- and NMN-degrading enzyme CD38 is elevated in the eyelids of older adults.
- In mice, the elevation in CD38 is associated with oil gland dysfunction, contributing to dry eye pathology.
- NMN enhances the effects of 78c, which inhibits CD38, in restoring oil gland function in aged mice.
From heart disease to dementia, many chronic diseases share the same underlying features, such as inflammation and biomolecule deficiency. What’s more, these underlying features seem to be perpetuated through a self-sustaining loop—a vicious cycle. For example, NAD+ (nicotinamide adenine dinucleotide) deficiency leads to inflammatory signals that recruit an enzyme called CD38. In turn, CD38 breaks down NAD+, exacerbating NAD+ deficiency and restarting the vicious cycle.
However, researchers from Kyoto University in Japan may have found a way to break the vicious cycle. As published in a new pre-print (not yet peer-reviewed), the researchers show that NMN enhances the effect of 78c in counteracting dry eye in aged mice, breaking the cycle of NAD+ deficiency, CD38 recruitment, and inflammation. The findings have implications for other chronic age-related diseases, like heart disease and dementia.
The Role of NAD+ In Dry Eye
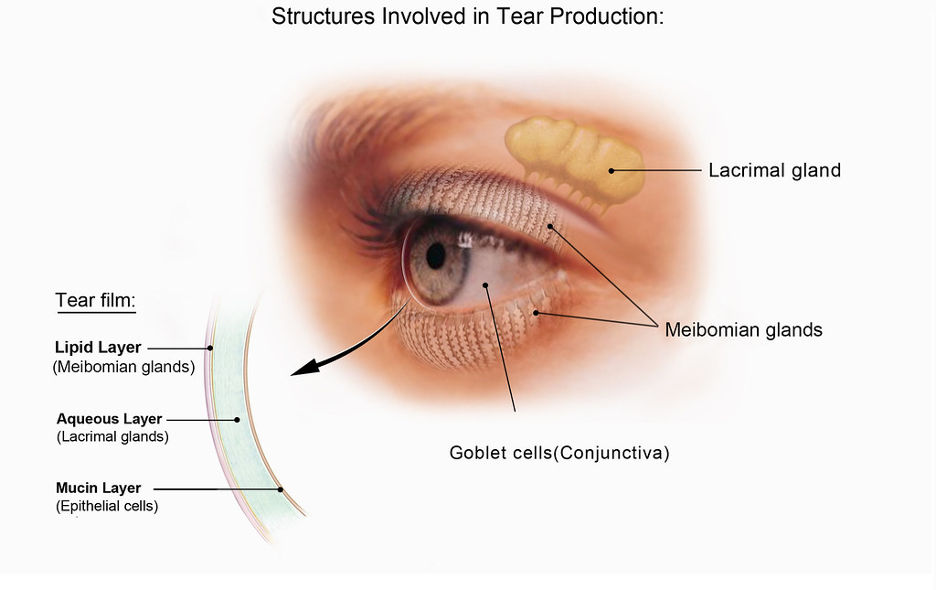
As a coenzyme, NAD+ allows enzymes to function. Among their vital roles, enzymes are responsible for synthesizing molecules that maintain cells. For example, an enzyme called Hsd3b6 (hydroxy-delta-5-steroid dehydrogenase, 3 beta- and steroid delta-isomerase 6) synthesizes hormones that maintain the function of oil gland cells within the eyelid. The Hsd3b6 enzyme needs NAD+ as a coenzyme to synthesize these hormones, which, in this case, promote the production of oil.
The oil glands within our eyelids are called meibomian glands, and their dysfunction is the most common cause of age-related dry eye. Demonstrating the critical role of hormone synthesis, the Kyoto University researchers previously recapitulated meibomian gland dysfunction in mice by genetically deleting the Hsd3b6 enzyme. It follows that, since NAD+ levels tend to decline with age, NAD+ deficiency could contribute to age-related dry eye by preventing hormone synthesis within meibomian cells.
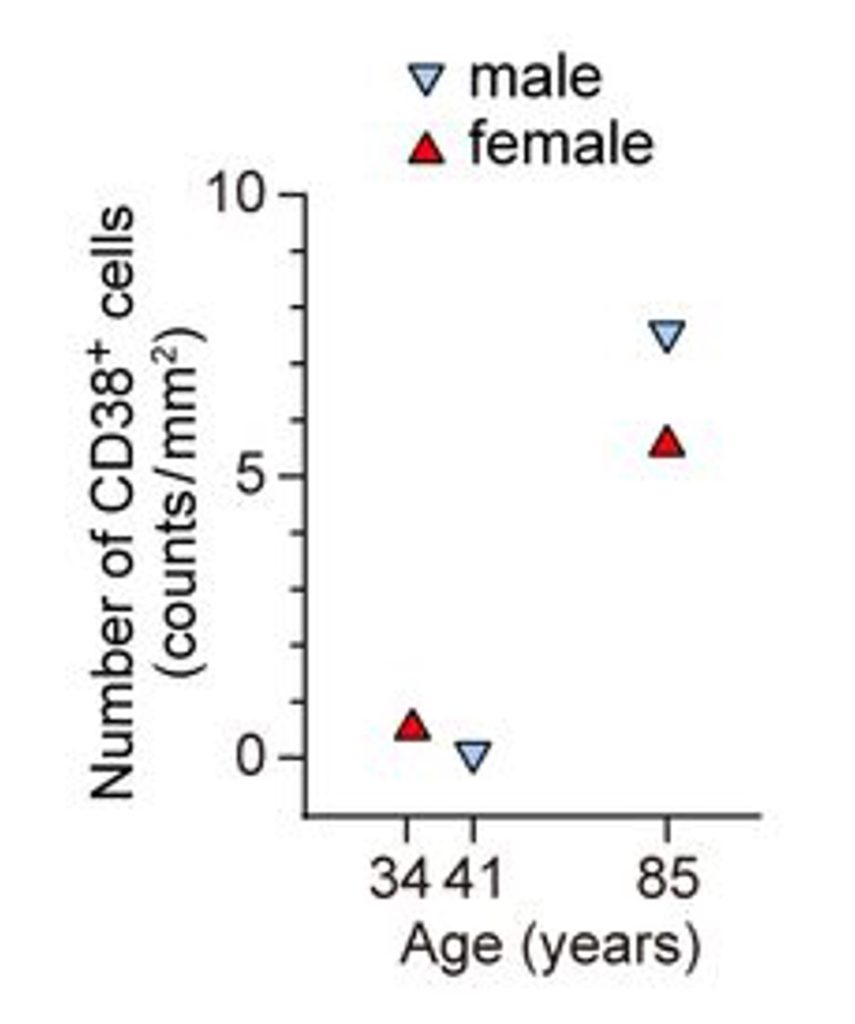
As part of their new study, the Kyoto University researchers found that impaired Hsd3b6 enzyme function was associated with elevated inflammation in mice. Crucially, the impairments in Hsd3b6 also increased CD38 levels in the eyelid. CD38 is an enzyme found on the surface of immune cells that breaks down NAD+ and NMN, causing lower NAD+ levels. In mice, it was shown that immune cells equipped with CD38 surrounded meibomian cells in response to inflammation. Similar results were observed in the eyelids of older adults, suggesting that CD38 could potentially influence dry eye in humans.
To test whether inhibiting CD38 can counteract eyelid aging, the Kyoto researchers injected 78c into the eyelids of aged mice. 78c is a small molecule known to inhibit CD38. It has been shown to prolong the lifespan of mice when injected into the bloodstream. Remarkably, 78c not only raised NAD+ levels but also counteracted meibomian gland atrophy. Furthermore, since CD38 breaks down NMN, the researchers injected the eyelids of mice with both NMN and 78c. This led to a greater elevation in NAD+ and enlargement of the meibomian glands, suggesting a potential synergistic effect.
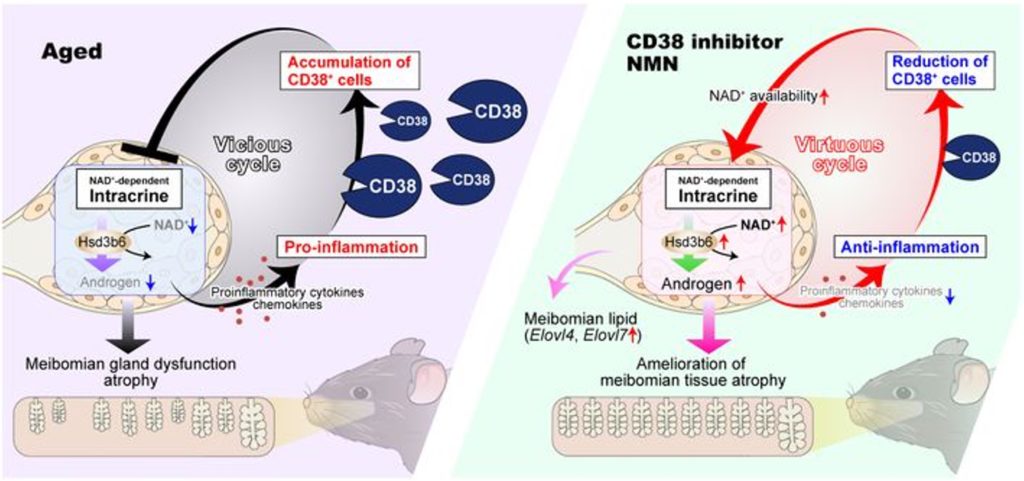
Together, the findings of the Kyoto University researchers suggest a new model for age-related dry eye. In this model, meibomian gland dysfunction occurs due to reduced hormone production from NAD+ coenzyme deficiency. This leads to an increase in inflammation and the recruitment of CD38 immune cells that reduce NAD+ levels and restart the vicious cycle. However, 78c and NMN replenish NAD+ levels, restore hormone production, counteract meibomian gland atrophy, prevent inflammation, and reduce CD38 activity, ending the vicious cycle.
The Aging Eye and Beyond
While the findings of the Kyoto University researchers are promising, there are still knowledge gaps that need to be filled. For example, it is unclear whether elevating NAD+ counteracts meibomian dysfunction by restoring Hasd3b6 enzyme activity. NAD+ is not just a coenzyme but a molecule that activates sirtuins, which are considered anti-aging enzymes. Supporting this, a previous study showed that NMN preserves eye cell health by activating sirtuins.
Furthermore, while the researchers did not directly measure dry eye, a previous study by the Kyoto researchers showed that the NAD+ precursors NMN or NR (nicotinamide riboside) alleviate dry eye in mice. Since inhibiting CD38 reduces NAD+ depletion and boosts NAD+ levels like NMN and NR, it can be assumed that a CD38 inhibitor can also alleviate dry eye. What’s more, combining an NAD+ precursor with a CD38 inhibitor may synergistically elevate NAD+ levels, although more studies are needed to confirm this.
Beyond the eye, boosting NAD+ counteracts numerous chronic diseases, such as heart disease and dementia, in animal models. Low NAD+ is linked to several biological drivers of aging, including mitochondrial dysfunction, DNA damage, and inflammation. Thus, replenishing NAD+ in cases of deficiency may counteract chronic diseases by targeting these drivers of aging. The results from human studies have been mixed, however, which could be due to varying levels of NAD+ deficiency in participants. Future studies that include participants with low NAD+ levels and exclude individuals with normal NAD+ levels may result in more robust anti-aging outcomes.
Model: 23-month-old male C57BL/6J mice
Dosage: 10 micrograms of 78c injected into the eyelid once every 3 days and 1 microliter of NMN dropped three times every 2 days for a total of 4 weeks

